Midweek Update

The American Hospital Association informs us that
The Senate early this morning approved on a party line vote a $3.5 trillion budget resolution, which included reconciliation instructions which will provide the majority party with the means to pass a comprehensive reconciliation package with just 51 votes in the Senate, rather than the usual 60-vote hurdle. The House will reconvene on Aug. 23 to consider the budget resolution. Once the resolution has passed both chambers, the House and Senate majorities can proceed with the reconciliation process, a resolution to which is expected in the fall.
Bloomberg adds
Translating the budget framework into law will require Biden and Democratic congressional leaders keeping their party’s moderate and progressive wings marching together.
Just hours after passage of the budget blueprint, Senator Joe Manchin, a Democrat from West Virginia, said he couldn’t support a social spending bill with a $3.5 trillion price tag. Senator Kyrsten Sinema, an Arizona Democrat, has said the same. One Democratic objection is all it would take to scuttle the package in the Senate.
Time will tell but we are talking about $3.5 trillion on top of the $1 trillion infrastructure bill and multi-trillion COVID-19 relief bills that Congress has passed in the last 18 months. It appears that Congress is trying to disprove the adage that money can’t solve all problems.
One of the initiatives in the budget reconciliation package is to add dental, vision, and hearing coverage to Medicare. Kaiser Health News discusses the issue here.
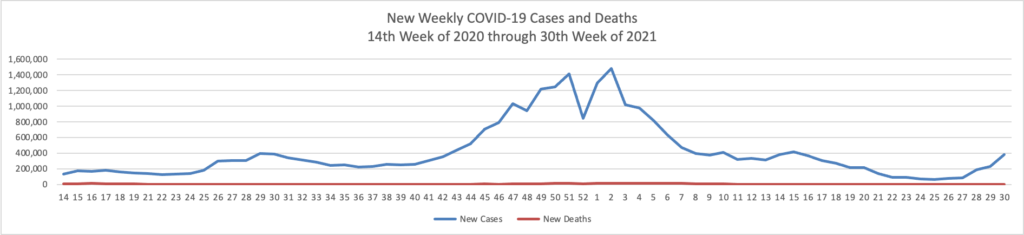
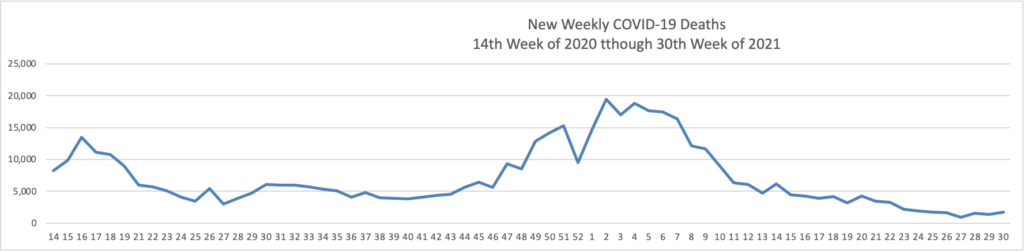
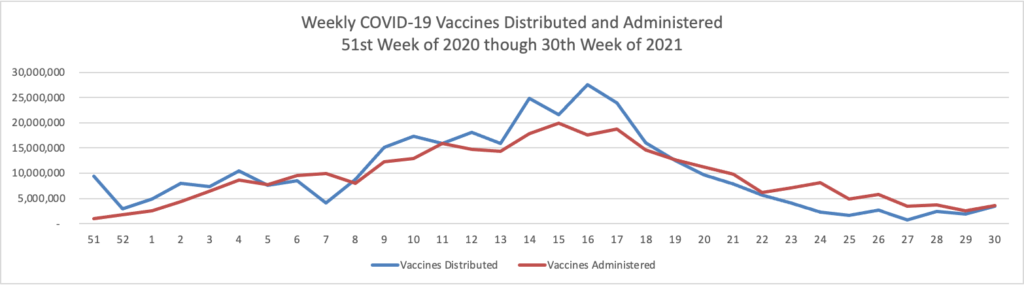
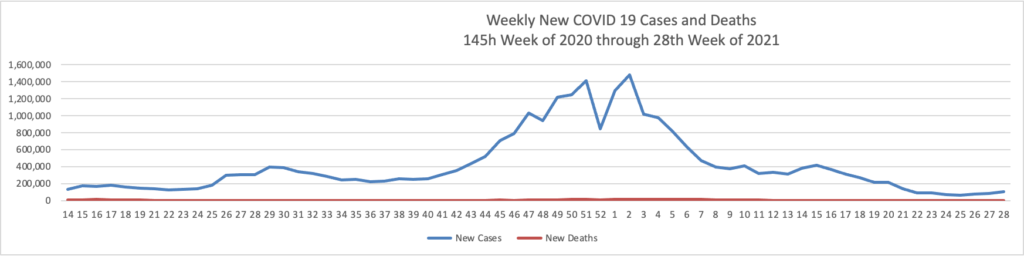
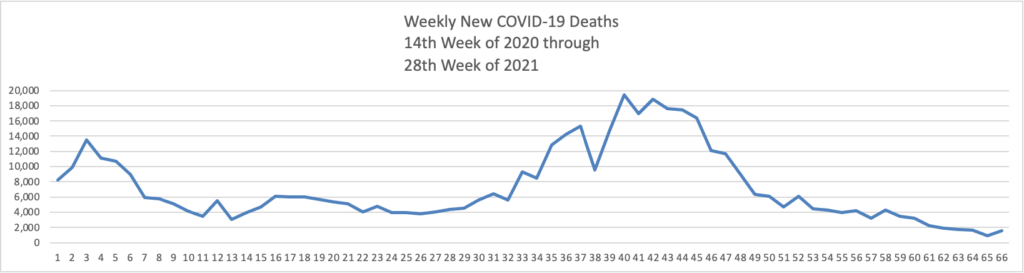
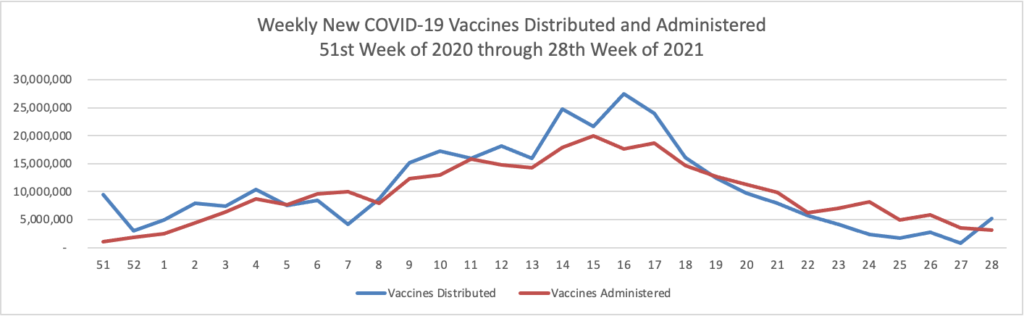
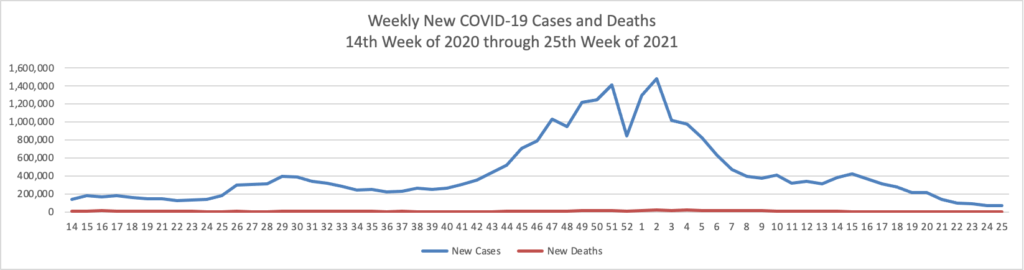
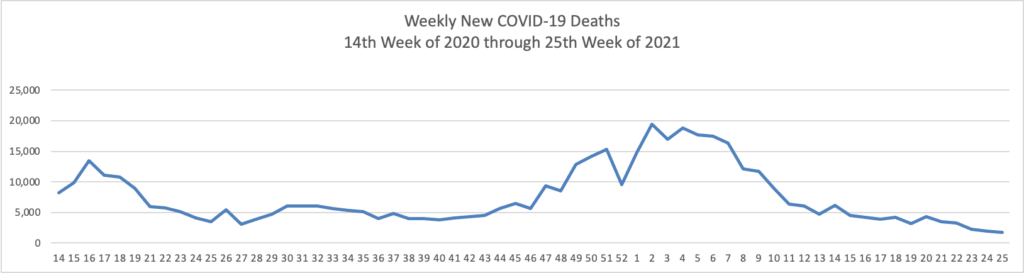
From the Delta variant front
- The Centers for Disease Control’s Advisory Committee on Immunization Practice released a helpful report on COVID-19 adverse side effects which it summarized as follows
What is already known about this topic?
Rare serious adverse events have been reported after COVID-19 vaccination, including Guillain-Barré syndrome (GBS) and thrombosis with thrombocytopenia syndrome (TTS) after Janssen COVID-19 vaccination and myocarditis after mRNA (Pfizer-BioNTech and Moderna) COVID-19 vaccination.
What is added by this report?
On July 22, 2021, the Advisory Committee on Immunization Practices reviewed updated benefit-risk analyses after Janssen and mRNA COVID-19 vaccination and concluded that the benefits outweigh the risks for rare serious adverse events after COVID-19 vaccination.
What are the implications for public health practice?
Continued COVID-19 vaccination will prevent COVID-19 morbidity and mortality far exceeding GBS, TTS, and myocarditis cases expected. Information about rare adverse events should be disseminated to providers, vaccine recipients, and the public.
- Forbes tells us that today “The Centers for Disease Control and Prevention encouraged anyone pregnant and breastfeeding to get vaccinated against coronavirus Wednesday, pointing to a growing amount of evidence that vaccines are safe and effective as new cases and hospitalizations linked to the virus surge across the country.” The Washington Post adds “Just 23 percent of pregnant women have received at least one shot of vaccine.”
- Also from Forbes as schools begin to reopen “Vaccine rates among teenagers have remained lower than the U.S. population overall, with only 43% of 12- to 15-year-olds and 52.8% of 16- and 17-year-olds receiving at least a first dose as compared with 58.9% of the total population and 71.2% of adults.”
- The Boston Globe discusses this teenage hesitancy issue — “The top reservation among parents of unvaccinated teens was the lack of information about the long-term effects of the shot, followed by concerns about side effects and fertility — despite conclusive evidence that the vaccine has no negative impacts on reproduction. Dr. Jill Kasper, a pediatrician at Cambridge Health Alliance, said she typically encounters ‘very little hesitancy’ from parents about routine adolescent vaccinations. That hasn’t been the case with COVID-19.”
The HHS Agency for Healthcare Quality and Research issued two noteworthy studies today. Here are the topline findings
- #1 Overuse or low-value procedures may result in patient physical, psychological, or emotional harm. This study explored the association between eight low-value care procedures and length of stay (LOS) and cost. All eight procedures were associated with increased LOS and cost, particularly spinal fusion. Patients receiving low-value care may be exposed to increased risk of adverse events and hospital-acquired conditions.
- #2 Medication administration errors made by parent or caregivers can result in medication errors at home. This systematic review found that 30% to 80% of pediatric patients experience a medication error at home, and that the risk increases based on characteristics of the caregiver and if a prescription contains more than two drugs.
Healthcare Dive informs us that
- CVS Health said its Aetna unit will offer virtual primary care to self-funded employers nationwide in a move that underscores the growing popularity of telehealth services fueled by the COVID-19 public health emergency.
- Using Teladoc Health’s physician-led care team model, the Aetna Virtual Primary Care service is intended to help strengthen the patient-doctor relationship and improve access to care, the vertically integrated company announced Tuesday. Members can receive health services remotely and in person.
Such support for primary care should be applauded.
In other healthcare news
- STAT News reports that The Department of Veterans Affairs has decided not to cover a new Alzheimer’s drug from Biogen [Adulhelm], citing insufficient evidence of “a robust and meaningful clinical benefit” and concerns about safety. In a notice issued by the agency, the VA said it will make exceptions for “highly selected patients” and listed several hurdles that must be cleared before the medicine will be covered, such as requiring it to be prescribed by providers who specialize in treating dementia and ensuring patients had recently received MRI brain scans. * * * ‘Its bad news for Biogen,’ said Ira Loss of Washington Analysis, which tracks legislative and regulatory issues affecting the pharmaceutical industry for investors, who noted several private insurers have either declined or delayed coverage for the drug. ‘The VA is a big buyer of medicines. And you don’t like this kind of publicity, that’s for sure.’”
- Fierce Healthcare continues to report from the HIMSS conference in Las Vegas. “Health IT giant Epic launched a new customer story-sharing website that lets Epic users share insights, tips and creative ideas around using health IT to improve their organizations and patient care. The site, EpicShare.org, combines insights from industry leaders, quick tips on improving outcomes and performance, in-depth case studies as well as a “Hey Judy” column from Epic founder and CEO Judy Faulkner.”