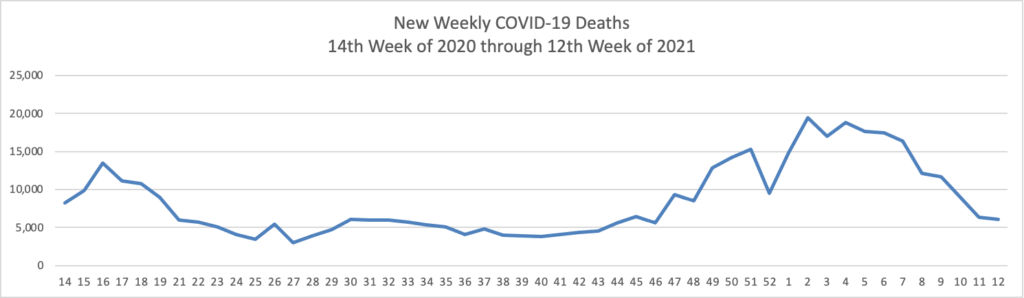
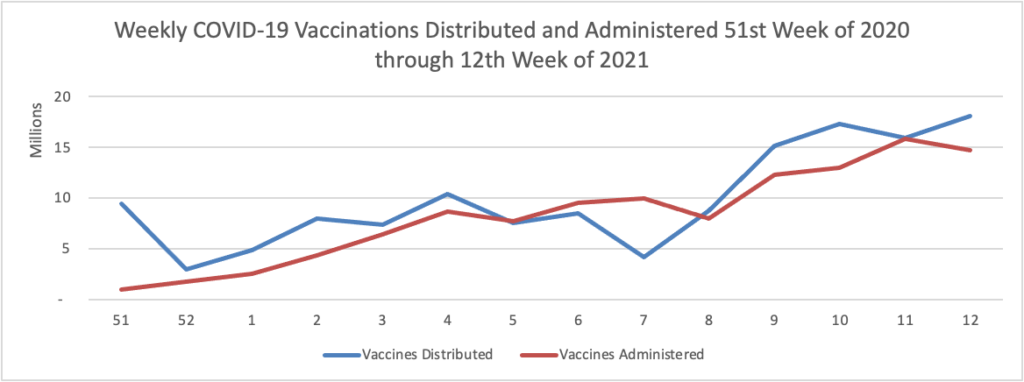
Thursday Miscellany

Yesterday, the Office of Personnel Management issued its technical guidance supporting the call letter for 2022 benefit and rate proposals which are due by May 31, 2021. The guidance provides insight into the call letter’s priorities.
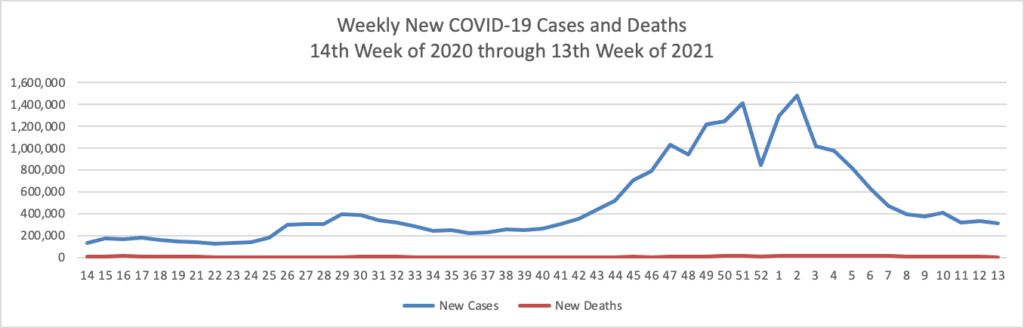
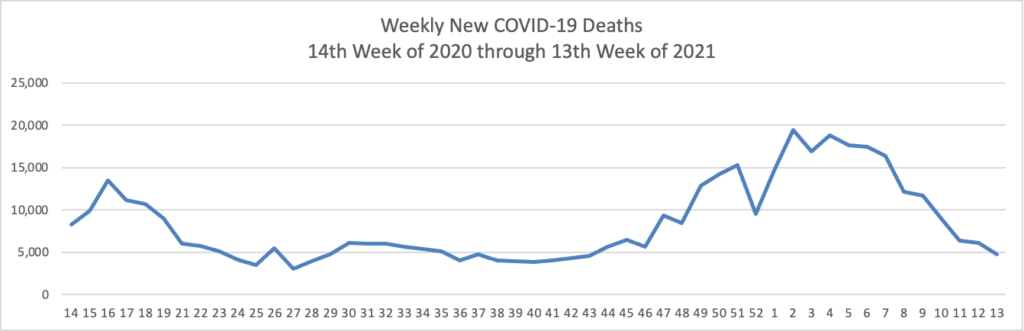
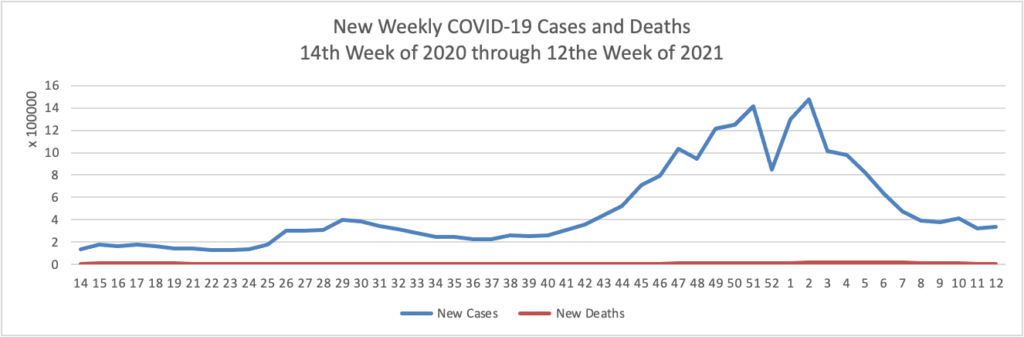
The FEHBlog’s priority is supporting the COVID-19 vaccination effort. STAT News reports that
Useful Covid-19 information isn’t reaching the Instagram generation [Z]. There’s almost no messaging specifically tailored to them from federal or state public health officials. There’s hardly anything official on Tik Tok. And even the limited efforts to reach them where they are — like Instagram’s links to its “Covid-19 information center”— aren’t working. * * * Numerous public health officials told STAT that the issue of growing vaccine reluctance among young people can be solved with a coordinated campaign of reliable, useful information that makes it both easy and enticing for young people to get vaccinated, even if they may not personally benefit much. Those same officials acknowledged, however, that much of the groundwork for messaging to young people is yet to be done.
Recognizing a problem is the first step toward solving it.
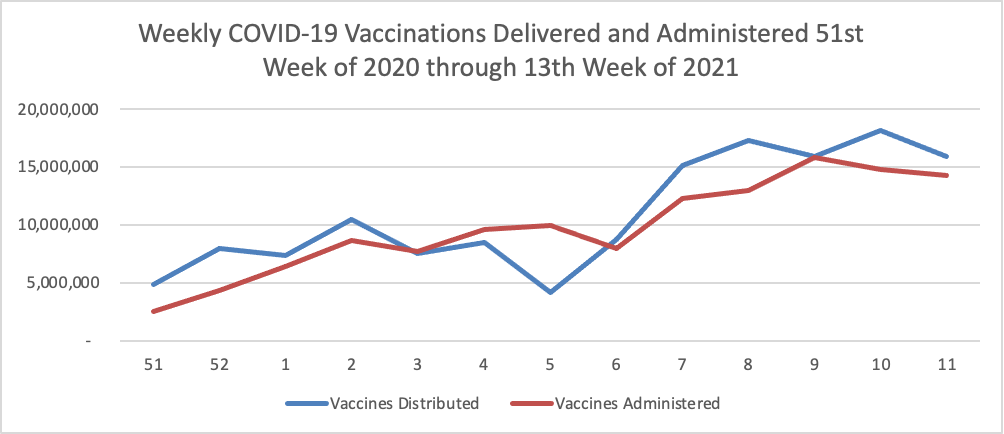
Govexec.com informs us that
FEMA has sent about 3,000 employees to vaccination sites and is fully running 30 mass vaccination centers. It declined to say how many volunteers it was seeking from other agencies. The emergency response agency is standing up community vaccination centers fully run and staffed by federal employees, providing funding and staffing to state-run sites and deploying mobile vaccination centers.
Thanks Feds.
The Centers for Medicare and Medicaid Services announced yesterday a proposed rule “that would update Medicare payment policies and rates for the Inpatient Psychiatric Facility Prospective Payment System (IPF PPS) for Fiscal Year (FY) 2022 and propose changes to the IPF Quality Reporting (IPFQR) Program. * * * Total estimated payments to IPFs are estimated to increase by 2.3% or $90 million in FY 2022 relative to IPF payments in FY 2021. For FY 2022, CMS is proposing to update the IPF PPS payment rates by 2.1% based on the proposed IPF market basket update of 2.3%, less a 0.2 percentage point productivity adjustment.”
Fierce Healthcare alerts us that “Drugs for inflammatory autoimmune conditions account for a growing chunk of pharmacy spend, according to new data from Prime Therapeutics. The pharmacy benefit manager studied (PDF) data on its 15 million commercially insured members in 2019 and 2020 and found that fewer than 1% of members had an inflammatory autoimmune condition such as psoriasis, rheumatoid arthritis, ulcerative colitis or Crohn’s disease. However, drugs treating these conditions accounted for nearly 20% of drug spend in the medical and pharmacy benefit, Prime Therapeutics found.” Wow.
Healthcare Dive reports that
- While urgent care centers do keep some lower-acuity patients from visiting costly emergency departments, their presence is not associated with a drop in total healthcare costs, according to a report in Health Affairs.
- Using commercial claims and enrollment data from 2008 to 2019, researchers found an increase of 1,000 lower-acuity urgent care visits resulted in 27 fewer lower-acuity ER visits. The entry of a high-volume urgent care center into a ZIP code was associated with a 31% drop in emergency visits.
- However, while ER trips were far more costly ($1,716 vs. $178), each $1,646 ER visit was offset by $6,327 more in urgent care costs because the number of urgent care visits per enrollee required to reduce one ER visit was 37.
Hmmm.
Health Payer Intelligence discusses a three ways for employers to fund their employee health benefit plans — insured, self-funded and an approach (with which the FEHBlog was not familiar) level funding.
In the level-funded model, the employer pays the insurer each month to cover expected healthcare expenditures, the Society for Human Resource Management (SHRM) website explains. The funds go towards claim payments, stop-loss insurance premiums, and administrative costs.
If this sounds familiar that is because this model borrows from both the fully-insured and self-insured models.
However, the distinction is that in a level-funded health plan, the insurer will return to the employer any funds that remain at the end of the year, if the volume of medical claims is not as high as anticipated. Alternatively, if the volume of medical claims exceeds the projected cost, employers will face a higher stop-loss insurance premium.
Although this is the general template for a level-funded plan, contracts may include various specifications, the SHRM site says. For example, some insurers may require that their company retain a certain percentage of the savings or that these funds roll over to be spent on medical claims in the subsequent year.
In this model, insurers—specifically larger insurers—may have cemented their level-funded plan offerings or they may work with employers to tailor the funding plan to fit the business’s needs.
Level-funded plans cater to smaller firms that want a self-insured health plan but may not be able to afford the high cost of medical claims and stop-loss insurance.
You do learn something new everyday.