Thursday Miscellany

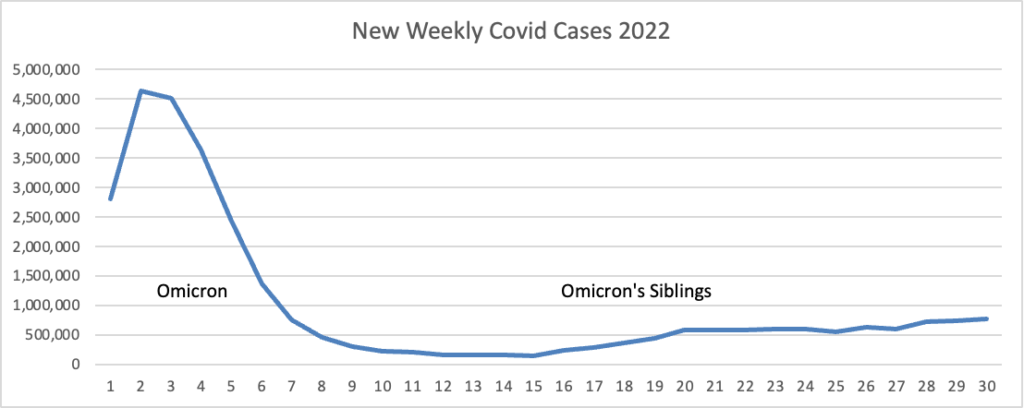
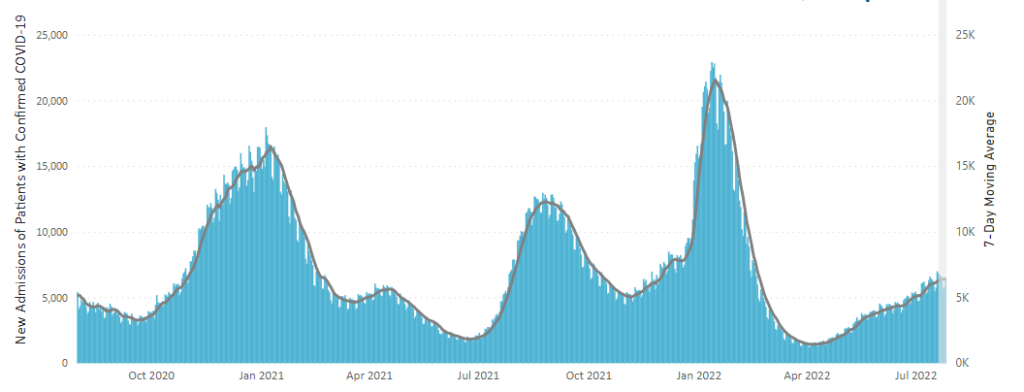
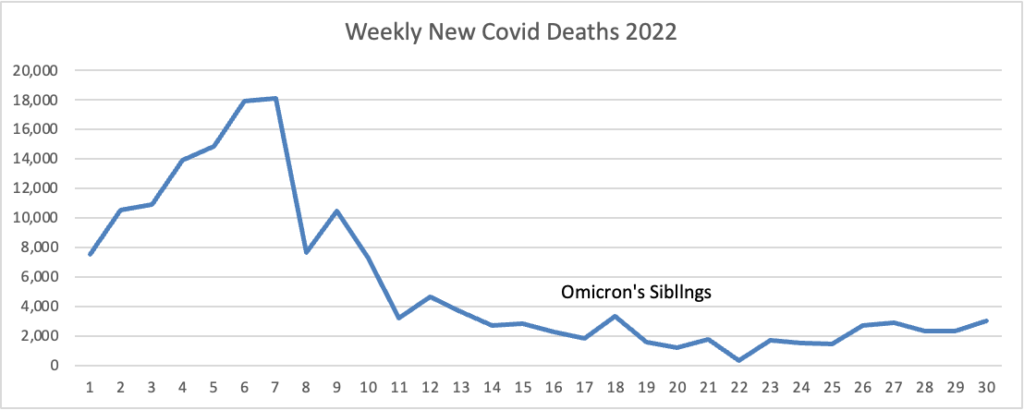
From the omicron and siblings front, MedPage Today provides us with good news.
Older patients treated with nirmatrelvir boosted by ritonavir (Paxlovid) for COVID-19 had lower rates of hospitalization and death compared with those not treated with the antiviral during the Omicron wave, according to an observational retrospective cohort study from Israel.
Among patients ages 65 and older, the rate of hospitalization due to COVID was 14.7 cases per 100,000 person-days for the 2,484 patients who received nirmatrelvir compared with 58.9 cases per 100,000 person-days for the 40,337 untreated patients (adjusted HR 0.27, 95% CI 0.15-0.49), reported Ronen Arbel, PhD, of Clalit Health Services in Tel Aviv, and colleagues.
Death occurred in two nirmatrelvir-treated patients and in 158 untreated patients (aHR 0.21, 95% CI 0.05-0.82), they said in the New England Journal of Medicine.
Herd safety, indeed.
From the Rx research and development front, NBC News reports
Two doses of psilocybin pills, along with psychotherapy, helped people with alcohol use disorder reduce drinking for at least eight months after their first treatments, results from the largest clinical trial of its kind show.
During the eight-month trial, 93 men and women ages 25 to 65 were chosen to receive either two psilocybin doses or antihistamine pills, which the researchers used as a placebo. They all also participated in 12 psychotherapy sessions.
All of the volunteers were averaging seven alcoholic drinks at a time before the trial.
More than 80% of those who were given the psychedelic treatment had drastically reduced their drinking eight months after the study started, compared to just over 50% in the antihistamine control group, according to results published Wednesday in JAMA Psychiatry. At the end of the trial, half of those who received psilocybin had quit drinking altogether, compared to about one-quarter of those who were given the antihistamine.
STAT News reports
A vaccine Pfizer is developing protected older adults against the respiratory syncytial virus, or RSV, which is a common cause of hospitalization and death in the U.S., the company said Thursday.
The experimental vaccine, known as RSVpreF, is considered a key product in Pfizer’s pipeline of experimental drugs. Right now drug companies are close to launching several different products against RSV, focused on protecting both older adults and infants.
BioPharma Dive tells us
BioMarin, a California-based biotechnology company, said Wednesday that its gene therapy for hemophilia has been cleared for market by European regulators, marking a first-of-its-kind approval.
The therapy, known as Roctavian, was given conditional marketing authorization as a treatment for certain patients with hemophilia A, the more common version of the rare bleeding disorder. Specifically, Roctavian is to be used in adults with “severe” disease — hallmarked by exceedingly low levels of a blood-clotting protein called Factor VIII — who don’t have a history of developing antibodies that attack this protein.
With approval in hand, BioMarin is now working to secure reimbursement across the European Commission’s various member states. Jeff Ajer, the company’s chief commercial officer, said on a conference call Thursday that the plan is to immediately launch Roctavian in Germany, followed by France. The company expects Roctavian’s list price in Europe to be “around” 1.5 million euros, or roughly $1.5 million, net of all discounts, he said.
Ajer added that BioMarin expects to disclose the specific European list price in October, a number that will be in-line but lower than the comparable net price in the U.S.
From the mental healthcare front —
Fierce Healthcare tells us
Employers view long-term mental health as the key healthcare issue coming out of COVID-19, according to a new survey.
Nearly half (44%) of employers surveyed by the Business Group on Health said they have seen this trend in their workforces, and another 44% expect to see worsening mental health in the future. Most (85%) said they anticipate enhanced mental health benefits launched under the pandemic to continue.
Mental health conditions also ranked sixth on the survey’s list of conditions driving healthcare costs, with 17% of those surveyed saying it was a major driver. This is a marked increase from 9% in 2020 and 14% in 2021.
Brenna Shebel, vice president of the Business Group on Health, said during a briefing with reports Tuesday that many employers are tackling mental health challenges “at all different angles.”
Forbes adds
AHIP, which stands for America’s Health Insurance Plans, issued a statement and “advocacy vision” at a time its member health plans are looking beyond paying and coordinating just medical care but also integrating behavioral health care into benefit plans for their government, commercial and employer clients. * * * Further details of the policy proposals and commitments can be found here.
From the U.S. healthcare business front, we learn about financial results for two health systems involved with the FEHB Program.
Becker’s Hospital Review reports that ” Intermountain Healthcare [based in Utah and serves surrounding states] saw its revenues increase in the first half of this year, and its merger with SCL Health fueled a significant increase in net income, according to recently released financial documents. The financial documents are the first Intermountain has filed since completing a merger with Broomfield, Colo.-based SCL Health in April.
Beckers Hospital CFO Report tells us, “UPMC reported higher revenue in the first half of this year than in the same period of 2021, but the Pittsburgh-based health system’s operating income declined year over year, according to financial documents released Aug. 23. * * * “Throughout 2022, the continued effect of COVID-19, along with conditions in the labor and supply markets have resulted in cost growth in employment, staffing and other operating expenses in excess of revenue growth,” UPMC management wrote in the financial filing.”
Finally STAT News delves into why so many large healthcare companies are interested in Signify Health. Quite simply,
The bidding war over Signify Health — a health technology business that could fetch multibillion-dollar offers from Amazon, CVS, and UnitedHealth Group — is not about its dazzling software or a blockbuster AI algorithm.
The crush of corporate interest, experts said, stems from something much bigger: the opportunity to move medical services back into the home. In-home care is quickly becoming the biggest battlefield in America’s biggest business, with a huge array of companies seeking to move health care outside the institutional walls that have confined it for much of the last century.
Signify Health is among those at the forefront of the quest. The Texas-based company, founded in 2017 as the result of a merger, has built its business around delivering highly detailed assessments of patients by visiting with them in their bedrooms and living rooms, creating better visibility — and clearer data — about their lives and health status.