Busy Thursday

From Capitol Hill Roll Call reports
House Republicans are mulling an attempt to buy time for further negotiations on federal spending and deficits by passing one or more short-term suspensions of the statutory debt ceiling this summer, including potentially lining up the deadline with the end of the fiscal year Sept. 30.
No decisions on a cutoff date have been made, and it’s not yet clear when the Treasury Department will run out of cash to meet all U.S. financial obligations. But most analysts agree Congress will need to act at some point between early June and September, and lawmakers likely won’t want to leave the matter unaddressed before the August recess.
and
The Senate is taking its time getting to work for 2023.
Back in Washington after a two-and-a-half week recess, the chamber adjourned Thursday afternoon without adopting an organizing resolution, meaning committees will remain in their holdover state until at least next week.
Senate Majority Leader Charles E. Schumer announced the Democratic committee assignments for the new Congress, with Michigan Democratic Sen. Gary Peters, the Democratic Senatorial Campaign Committee chair, earning a coveted seat on the Appropriations Committee.
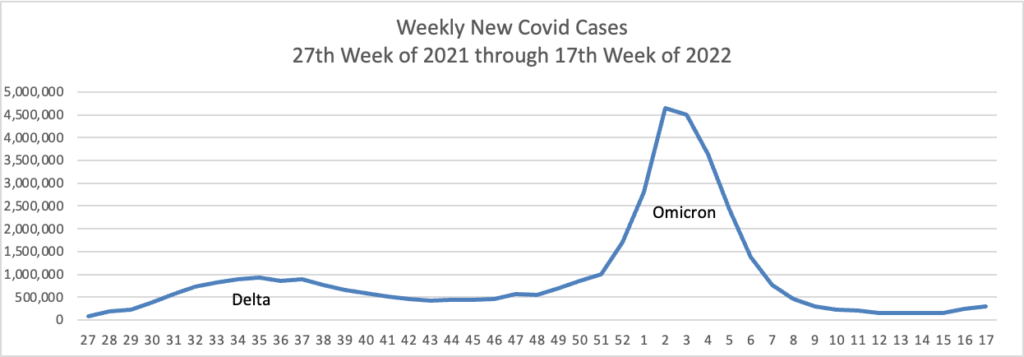
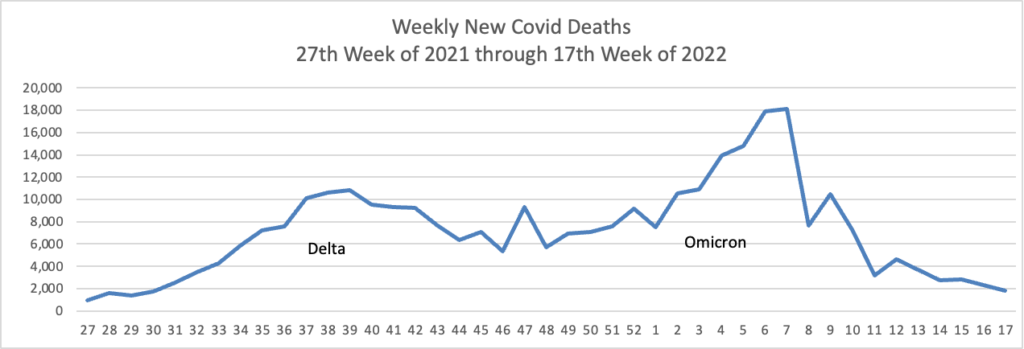
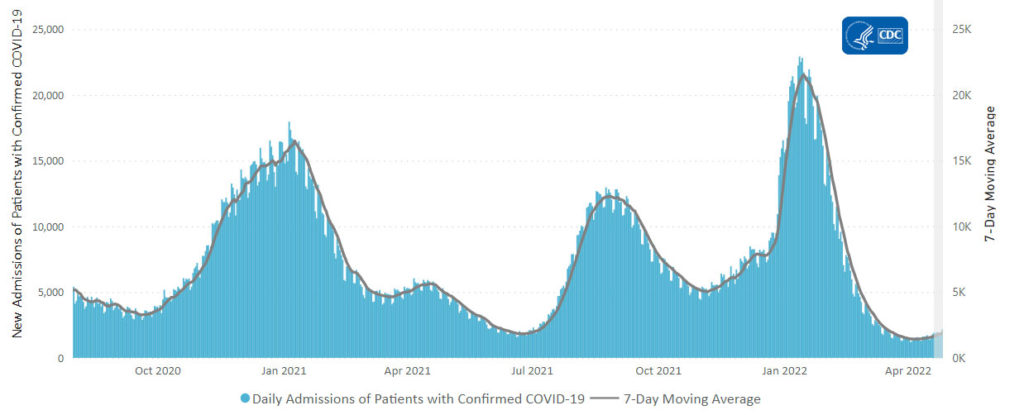
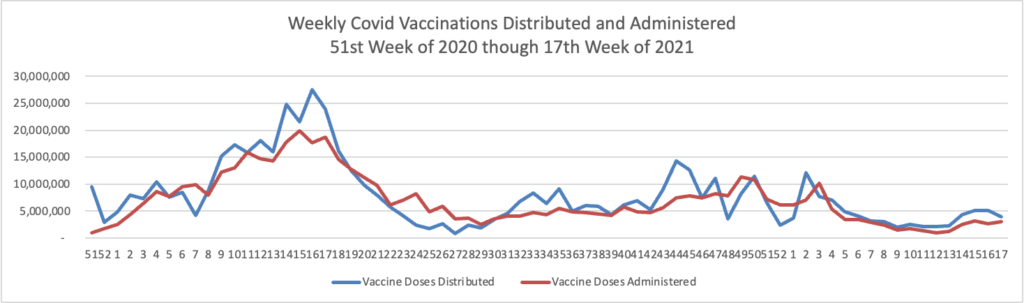
From the Omicron and sibligns front, The American Hospital Association tells us
A Food and Drug Administration Vaccines and Related Biological Products Advisory Committee [VRBAC] unanimously voted today to recommend harmonizing the composition of all primary series and booster doses administered in the U.S. For example, the composition of all vaccines administered going forward might be bivalent.
STAT News offers a complete report on today’s meeting. For example, STAT News explains
The FDA is also asking the members of VRBPAC their thoughts on its proposal that Americans get an annual Covid shot, in the way they get a flu shot, one that is reconstituted regularly to try to target the strains in circulation at the time. In documents the FDA made public before the meeting, it proposed choosing new vaccine strains in June for a vaccine campaign that would begin in September.
Covid is clearly here to stay, so this may sound sensible. But there are concerns some of this is still based on a leap of faith rather than a data-led process. For example, the idea that everyone might need an annual Covid booster will not earn a unanimous “yea” vote out of this expert panel.
The VRBAC recommendation is subject to FDA and CDC approval.
STAT News adds
The FDA on Thursday withdrew the authorization of Evusheld, the latest antibody therapy to be rendered ineffective by the mutations the virus has picked up. Notably, Evusheld — unlike other antibody therapies — was not for infected patients, but rather was given as a pre-exposure treatment to people at high risk for severe Covid-19, such as those with compromised immune systems.
In other FDA news
- The FDA announced, “Given the growing cannabidiol (CBD) products market, the FDA convened a high-level internal working group to explore potential regulatory pathways for CBD products. Today we are announcing that after careful review, the FDA has concluded that a new regulatory pathway for CBD is needed that balances individuals’ desire for access to CBD products with the regulatory oversight needed to manage risks. The agency is prepared to work with Congress on this matter. Today, we are also denying three citizen petitions that had asked the agency to conduct rulemaking to allow the marketing of CBD products as dietary supplements.”
- Fierce BioTech informs us “More than two years after submitting it for FDA review, Tidepool has scored the agency’s clearance for a smartphone app that allows people with Type 1 diabetes to build their own closed-loop “artificial pancreas” system.”
From the obesity treatment front —
HealthDay discusses findings made by “Utah researchers who followed patients for up to 40 years after they had one of four types of weight-loss (bariatric) surgery.”
Weight-loss surgery can literally be a lifesaver, cutting death rates significantly during the course of a decades-long study
Death from all causes was 16% lower, while it was 29% lower for heart disease, 43% lower for cancer and 72% lower for diabetes
But there were some troubling findings: These patients were 83% more likely to die of liver disease and 2.4 times more likely to die by suicide, mostly seen in younger patients
STAT News provides a two minute long video explaining how the new obesity drugs work.
STAT News also describes an unusual alliance that has banded together to lobby Congress to repeal a provision in the Medicare Modernization Act of 2023 that prohibits Part D from covering obesity drugs. “Recent scientific advances, media coverage, and advocacy have helped raise the profile of the issue on Capitol Hill, said Jeanne Blankenship, the vice president for policy initiatives and advocacy at the Academy of Nutrition and Dietetics. ‘It’s becoming front and center. I think we can’t turn our backs on it any longer,’ Blankenship said.”
From the Rx coverage front, Beckers Hospital Review introduces us to the three PBMs that have partnered with the Mark Cuban Pharmacy.
From the HIPAA / electronic health records front —
- MedPage Today reports, “Unique Patient Identifier Funding Once Again Barred by Congress— Biden administration working on better patient matching instead.” The FEHBlog will never understand Congress’s intransigence here.
- Healthcare Dive tell us “Interoperability continues to improve among U.S. hospitals, but there’s still a ways to go, according to new government data. More than six in 10 hospitals electronically shared health information and integrated it into their electronic health records in 2021, up 51% since 2017, the Office of the National Coordinator released in a Thursday data brief. The availability and usage of electronic data received from outside sources at the point of care has also increased over the last four years, reaching 62% and 71% respectively in 2021.”
From the NIH research front, NIH calls attention to its research studies on the role of the placebo effect in healthcare treatments and the link between hydration and better aging.
From the miscellany department —
- Mercer Consulting “projects the 2024 inflation-adjusted amounts for health savings accounts (HSAs), high-deductible health plans (HDHPs) and excepted-benefit health reimbursement arrangements (HRAs) will rise significantly from 2023 levels.”
- Benefits Consultant Tammy Flanagan, writing in Govexec, discusses the categories of family members who are eligible and ineligible for FEHB coverage.
- HR Dive identifies five trends that will share HR this year.