Tuesday’s Tidbits

From Capitol Hill, Fierce Healthcare reports
New bipartisan legislation introduced in the Senate aims to empower the Federal Trade Commission (FTC) to crack down on pharmacy benefit manager practices such as spread pricing.
The legislation, introduced Tuesday, comes as the PBM industry faces other areas of reform, including a proposed rule to get rid of clawback fees PBMs can charge pharmacies after the drug is dispensed.
Lawmakers said federal agencies need more power, though, to rein in PBM practices.
From the No Surprises Act (“NSA”) front, AHIP and the Blue Cross Association inform us
A recent survey and analysis conducted by AHIP and the Blue Cross Blue Shield Association (BCBSA) found that in the first two months of 2022, the NSA prevented more than 2,000,000 potential surprise medical bills across all commercially insured patients. If only a fraction of these claims are ultimately disputed through IDR, it would still far exceed the government’s estimate. Should the trend hold, more than 12,000,000 surprise bills will be avoided in 2022 due to the NSA.
The law is working to protect millions of consumers from costly surprise bills and yet several hospital and provider organizations have filed lawsuits challenging the NSA regulations and legislation in order to increase their own profits at patients’ expense. Recent polling conducted by Morning Consult on behalf of the Coalition Against Surprise Medical Billing found that 8 in 10 voters, after learning about the NSA, are concerned that lawsuits from physician and hospital organizations could delay or overturn the patient protections in the Act.
The findings of the AHIP-BCBSA survey are important to demonstrate how many consumers have already benefitted from the NSA and to underscore the extent of total claims that could be impacted if the IDR process is not a predictable process with payment amounts that trend towards market rates.
That’s great news.
In public health news, the American Hospital Association tells us ‘
U.S. births rose 1% in 2021 to about 3.7 million, the first increase since 2014, according to preliminary data released today by the Centers for Disease Control and Prevention. Birth rates declined for women aged 15-24 and rose for women aged 25-49. The cesarean delivery rate rose 0.3 percentage point to 32.1%, while the preterm birth rate rose 4% to 10.48%, the highest rate since 2007, CDC said.
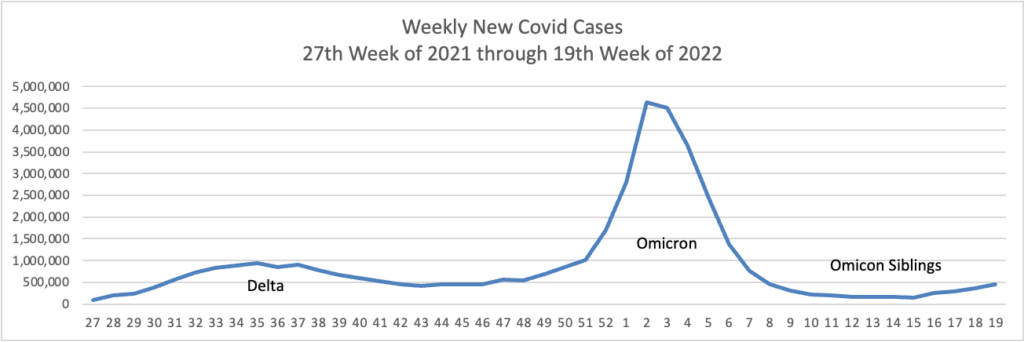
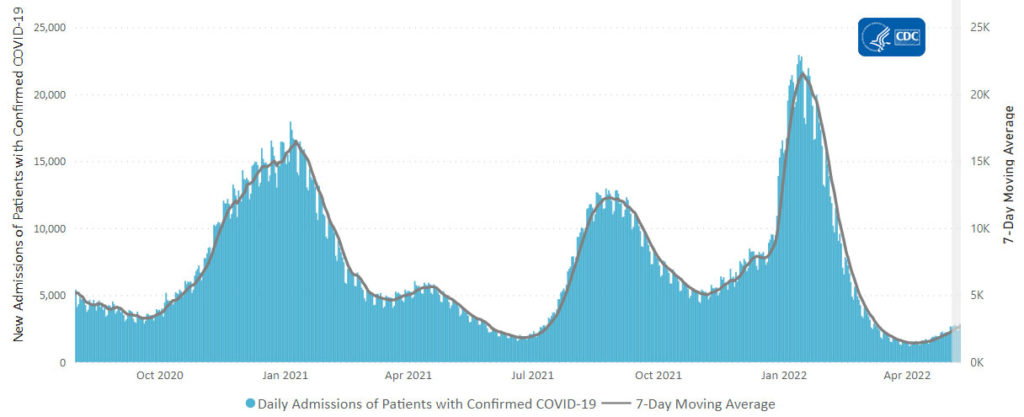
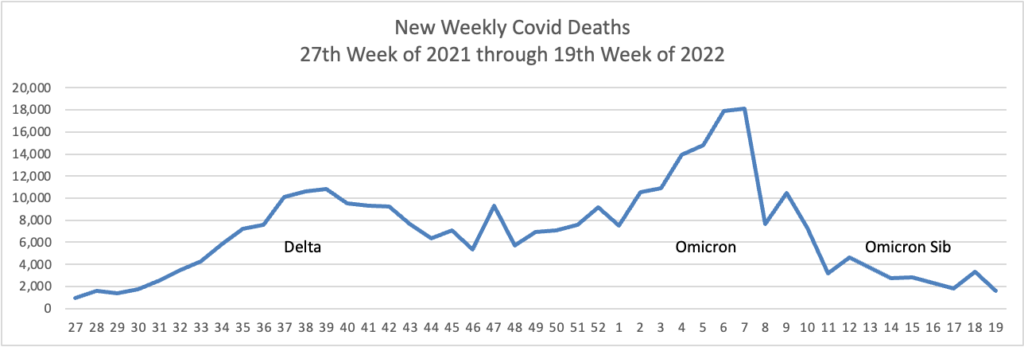
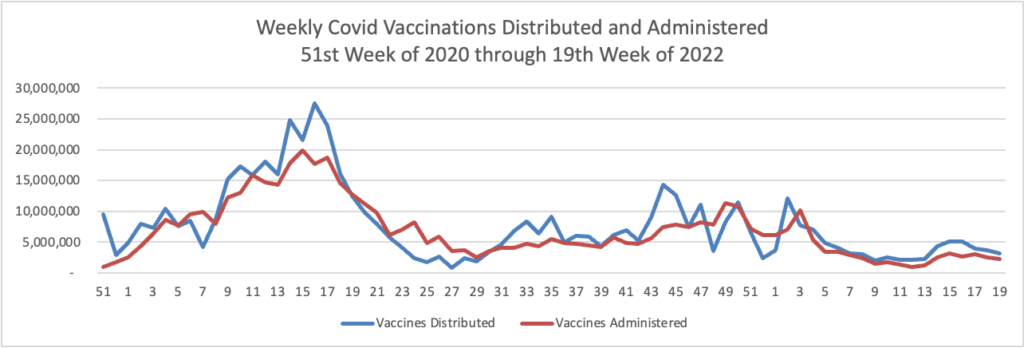
From the Omicron and siblings front
- Reuters reports “The U.S. Food and Drug Administration set June 14-15 as the new meeting date to review Moderna Inc’s emergency authorization request for its COVID-19 vaccine for children aged 6 months to 5 years and Pfizer Inc’s vaccine for those aged 6 months through 4 years.”
- Precision Vaccines tells us “The U.S. Centers for Disease Control and Prevention (CDC) issued today Health Alert Network Health Advisory CDCHAN-00467 to update healthcare providers, public health departments, and the public on the potential for recurrence of COVID-19 or “COVID-19 Rebound.” COVID-19 Rebound cases have been reported to occur between two and 8 days after initial recovery. They are characterized by a recurrence of COVID-19 symptoms or a new positive viral test after testing negative. A brief return of symptoms may be part of the natural history of SARS-CoV-2 (the beta coronavirus that causes COVID-19) infection in some persons, independent of treatment with Paxlovid and regardless of vaccination status.” STAT News offers a more detailed article on this topic for those interested.
In other virus news, the Hill informs us
Officials for the Centers for Disease Control and Prevention (CDC) on Monday said the agency is releasing doses of a smallpox vaccine in response to the few recent cases of monkeypox that have been detected in the U.S. Jennifer McQuiston, the deputy director for the CDC’s Division of High Consequence Pathogens and Pathology, said during a press briefing that more than a thousand doses of the Jynneos smallpox vaccine are currently available in the U.S., with more doses expected to become available as production ramps up. * * * The vaccines will be designated for people who are most likely to benefit from them, McQuiston said, including those who are known to have had close contact with monkeypox patients, health care workers and people who would be at high risk of developing a severe case of the disease.
From the healthcare business front, Beckers Hospital News identifies Walmart’s 18 Centers of Excellence in the U.S.
In National Institutes of Health news, NIH Reseach Matters discusses antioxidant effects on dementia risk and how the health benefits of dietary fibers depend on the fiber type, the amount, and the individual.”