Monday Roundup

The Senate’s bipartisan infrastructure bill cleared a cloture vote last night. The Senate is expected to pass it by tomorrow. Roll Call reports on the $3.5 trillion “human infrastructure” related budget reconciliation bill that the Congressional Democrat leadership unveiled today.
In Delta variant news
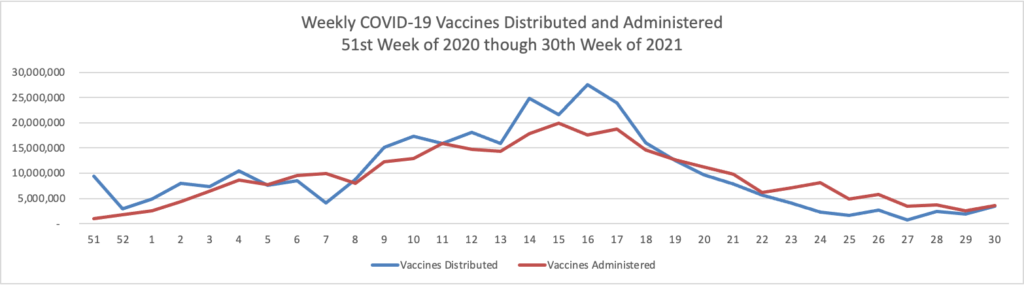
- Federal News Network informs us that “The Defense Department is taking actions to make COVID-19 vaccines mandatory for service members by mid-September, or once the vaccines gets licensure from the Food and Drug Administration, whichever comes first.”
- The New York Times reports that long COVID is now afflicting children. “Studies estimate long Covid may affect between 10 percent and 30 percent of adults infected with the coronavirus. Estimates from the handful of studies of children so far range widely. At an April congressional hearing, Dr. Francis Collins, director of the National Institutes of Health, cited one study suggesting that between 11 percent and 15 percent of infected youths might ‘end up with this long-term consequence, which can be pretty devastating in terms of things like school performance.’” Quite troubling. All the more reason to accelerate vaccinations for children.
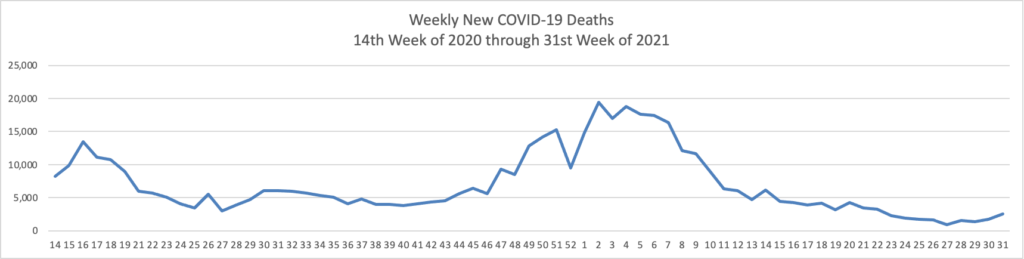
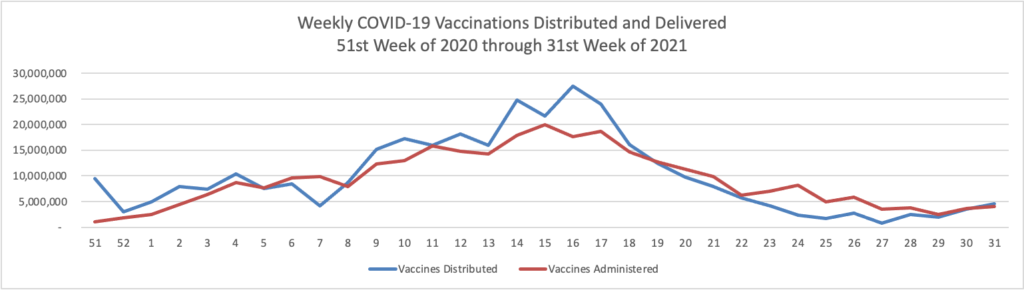
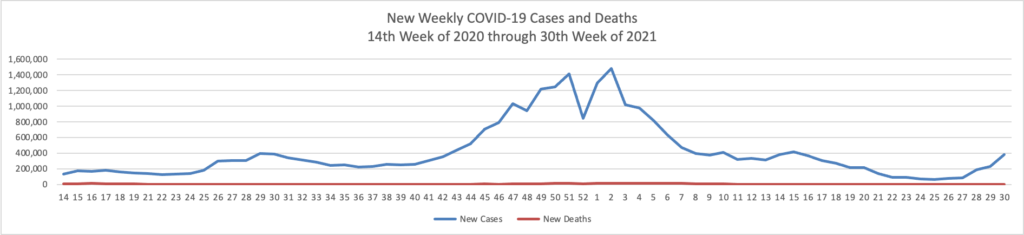
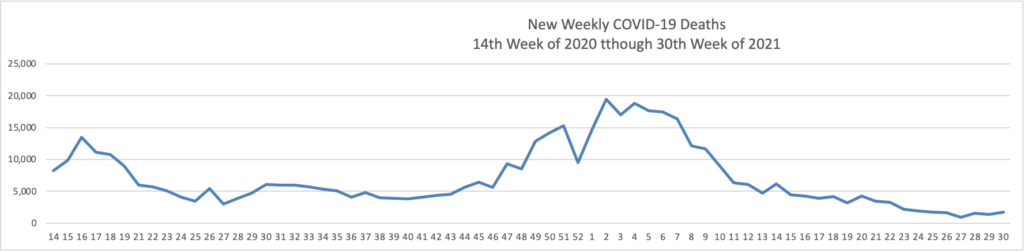
- In this regard, the Wall Street Journal reports that “As the highly contagious Delta strain tears through the country, the trends thus far suggest vaccines can turn Covid-19 into a less dangerous, more manageable disease. “Vaccines definitely make a difference,” said David Dowdy, an epidemiologist at the Johns Hopkins Bloomberg School of Public Health.A Wall Street Journal analysis shows sharp geographic divides in vaccination and hospitalization levels, with every state that has an above-average vaccine rate showing below-average hospitalizations, including in well-vaccinated New England. In the South, meanwhile, fewer people are vaccinated on average and hospitalization rates are climbing faster.” Lesson hopefully learned.
From the second quarter financials report front, Fierce Healthcare tells us that “Kaiser Permanente reported $3 billion in net income for the second quarter of the year as membership in its health plan remains steady. The health system and insurer posted total operating revenues of $23.7 billion against total operating expenses of $23.3 billion. The revenue was slightly above the $22.1 billion it earned in the second quarter of 2020. Kaiser noted in its earnings statement Friday that favorable financial market conditions resulted in $3 billion in net income, compared with $4.5 billion for the second quarter of 2020.”
From the No Surprises Act front, a friend of the FEHBlog pointed out to him that a second NSA rule has been pending at the Office of Information and Regulatory Affairs (“OIRA”) for the past month:
AGENCY: HHS-CMS RIN: 0938-AU61 Status: Pending Review
TITLE: Reporting Requirements Related To Air Ambulance and Agent and Broker Services and HHS Enforcement Provisions (CMS-9907)
STAGE: Proposed Rule ECONOMICALLY SIGNIFICANT: Yes
RECEIVED DATE: 07/07/2021 LEGAL DEADLINE: Statutory
In other news —
- Health Day tells about a severe blood shortage that is adversely affecting U.S. hospitals. “‘A blood shortage is when we have demand outpacing our supply,’ noted Paul Sullivan, senior vice president of donor services for the American Red Cross. ‘Usually it’s around some challenging period of the year. The beginning of summer, end-of-year holidays. And obviously we work hard to try to plan for those times.’ But this time is different, Sullivan said, with the COVID-19 pandemic and its fallout triggering a huge spike in hospital demands for blood. The result is a critical blood shortage that’s now seven weeks long and counting.” The article concludes “There’s more on the blood shortage and blood donations at the American Red Cross.”
- In good news, Health Leaders Media reports that “The years-long effort to reduce unnecessary Cesarean section births in the United States is coming to fruition, an obstetrics expert says. Complications from C-sections such as hemorrhaging are widely considered to be a contributing factor to the country’s high maternal mortality rate. The federal Centers for Disease Control and Prevention have been monitoring maternal mortality since 1986. The number of pregnancy-related deaths has risen steadily since the monitoring effort began, from 7.2 deaths per 100,000 live births in 1987 to 18.0 deaths per 100,000 live births in 2014. “We are finally at the point where most hospitals are sharing their data and having conversations on an individual basis about C-section rates. We are having conversations about quality at labor and delivery units as well as about the quality of individual providers. It has taken more than a decade to get to this point,” says Amy VanBlaricom, MD, vice president of clinical operations for western states at Greenville, South Carolina-based Ob Hospitalist Group.”