Monday Musings
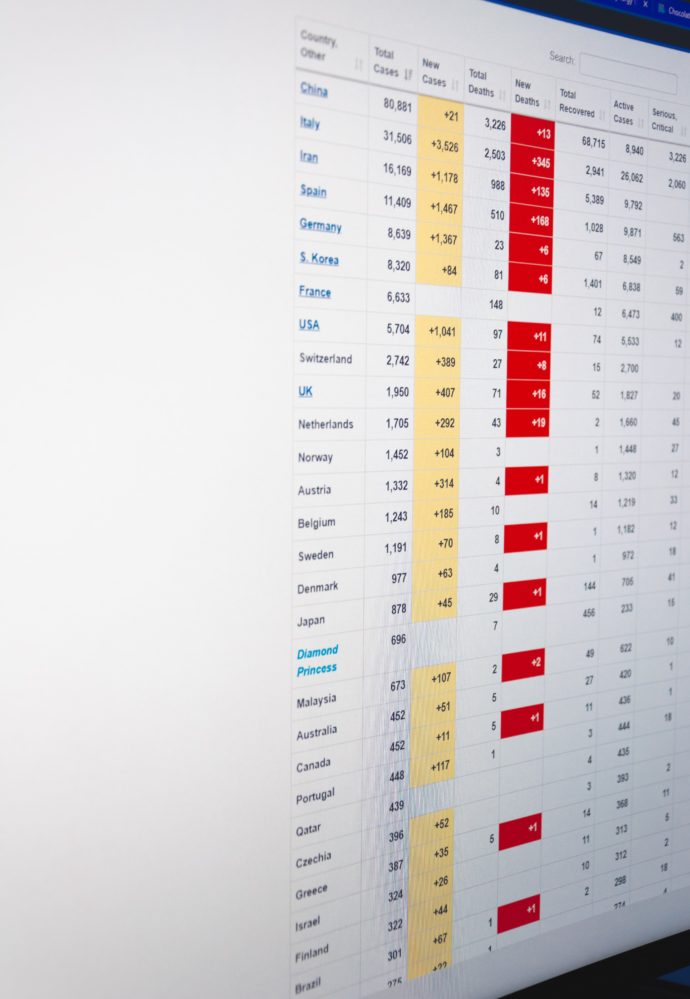
Becker’s Hospital Review provides a helpful update of today’s top seven COVID-19 news items.
Healthleaders Media interviews “Karen Murphy, PhD, RN, executive vice president and chief innovation officer at Geisinger, and founding director of the Steele Institute” for Health Innovation about COVID-19 and lessons for the future.
HL: Is telehealth among the initiatives that should be expanded? What else?
Murphy: Virtual care is one thing that I think will transform the healthcare industry. And, we’re looking at what we need in terms of infection control. How do we change our practices?
Federal News Network reports on legislative ideas coming from the majority leadership of the House Oversight and Reform Committee. With regard to the FEHBP, the leaders
recommended allowing federal employees to change or opt into new insurance plans during a health emergency like the current pandemic.
Currently, FEHB participants have one chance a year, usually a four-week “open season,” to make changes to their health insurance plan.
In addition, the chairmen suggested allowing employees to extend eligibility for their federal health, dental and vision benefits to qualifying dependents for an additional year. Some children, for example, may age out of their family’s current health plans during the coronavirus pandemic, they said.
The FEHBlog recalls that approximately 15% of FEHBP eligible employees decline FEHBP coverage in favor of taking coverage under his or her spouse’s employer sponsored coverage. We the rapid employment loss due to the great hunkering down, it would make sense for OPM to remind federal employees that losing other health coverage is a qualifying life event that allows them to rejoin the FEHBP for themselves and their eligible family members. OPM prominently notes that “Generally, if you choose to make a change to your enrollment, you must make the change within 60 days of the event.”
Today, the U.S. Supreme Court, which had cancelled its March and April oral arguments, rescheduled ten of those cases for telephonic oral argument in May. “The Court anticipates providing a live audio feed of these arguments to news media.” Normally the press desiring live feeds would have listen to the oral argument in person at the Court. Transcripts of the oral argument come later.