Thursday Miscellany

From Capitol Hill, the Hill reports
The Senate has locked in a deal to quickly pass a massive government funding bill that includes $13.6 billion in Ukraine aid.
The agreement, announced by Senate Majority Leader Charles Schumer (D-N.Y.), puts the funding bill on a glide path to pass on Thursday night, capping off hours of would-they-won’t-they drama.
Mazaal tov to Congress.
Also on Capitol Hill today, the Senate Homeland Security and Governmental Affairs Committee held a confirmation hearing for Krista Boyd, the President’s nominee to serve as OPM Inspector General. Fedweek notes that “Ms. Boyd is a senior staff member of the House Oversight and Reform Committee with long experience on Capitol Hill in federal workplace matters.”
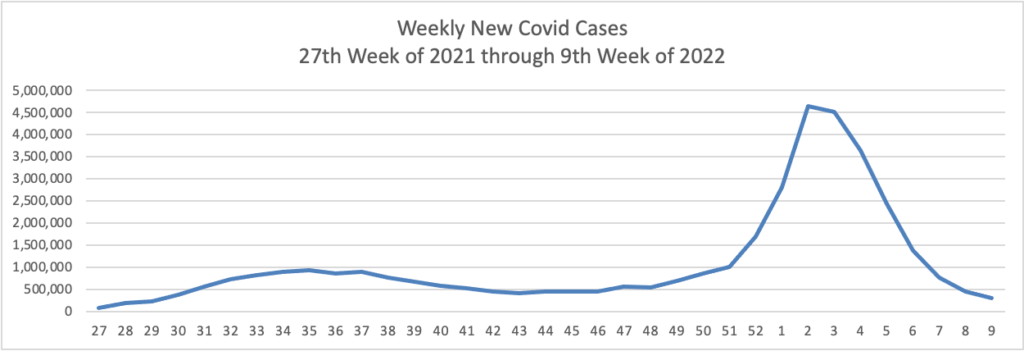
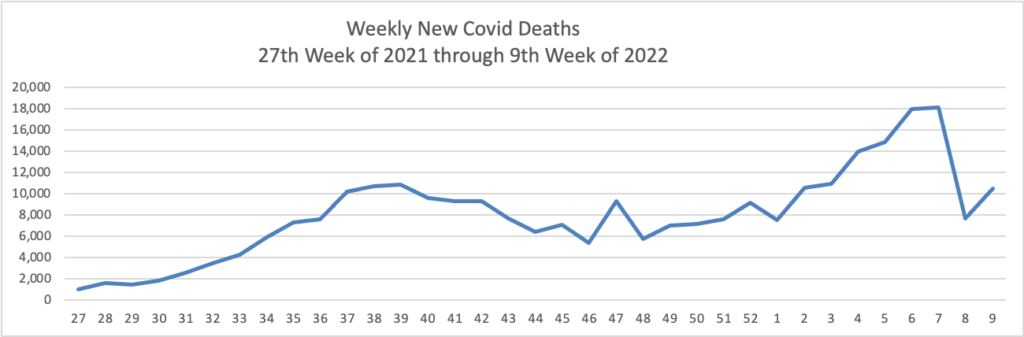
From the Omicron front, Becker’s Hospital Review informs us “The rate of new COVID-19 cases involving the omicron subvariant BA.2 appears to be slowing in the U.S., according to variant proportion estimates from the CDC.”
Also, the Justice Department announced “Effective immediately, Associate Deputy Attorney General Kevin Chambers will serve as the Director for COVID-19 Fraud Enforcement.”
From the litigation front, Reuters reports “The judge overseeing Purdue Pharma’s bankruptcy on Wednesday approved a $6 billion opioid settlement funded by its Sackler family owners, overruling objections from the Department of Justice and 20 states that opposed the deal.”
From the healthcare business front
Healthcare Dive tells us
Anthem plans to change its name to Elevance Health, if the move is approved by shareholders, the company said Thursday.
The new name is meant to reflect the company’s offerings beyond traditional health insurance. “Elevance Health’s companies will serve people across the entire care journey, connecting them to the care, support, and resources they need to lead healthy lives,” Anthem CEO Gail Boudreaux said in a statement.
Elevance was chosen as a combination of the words “elevate” and “advance.” There will not be any changes to leadership or organizational structure accompanying the new name.
If approved, the Elevance name will start being used at the end of the second quarter of this year. Anthem Blue Cross and Blue Shield plans will still use the Anthem name.
From the telehealth front, Healthcare Dive reports
[Virtual care vendor] Amwell and LG Electronics are teaming up to jointly develop new digital health devices and tools, starting with hospital care in the U.S., the companies announced Wednesday.
South Korea-based LG, which manufactures a wide range of devices from refrigerators to computer monitors, already provides smart TVs for inpatient rooms.
Now, through the partnership, LG will also create devices that can host services from Amwell’s virtual care platform, Converge.
PYMTS.com reports this electronic health records news from the Vive conference being held in Miami
Electronic health records containing some of the most guarded personal data about people are making headlines again as a consortium of players join forces to create a universal single sign-in, allowing patients secure access to unified health data via digital identity.
Coming out of the ViVE health technology conference happening this week in Miami Beach, the effort is led by consumer-directed healthcare advocacy group the CARIN Alliance, working together with the Department of Health and Human Services (HHS) and other stakeholders.
On Tuesday (Mar. 8), Politico reported that HHS “is working with several health systems, insurers and health tech groups to roll out a single way for patients to log in and access their medical records across multiple systems. The launch later this month will set up a test environment for integrating the technology, said Ryan Howells, principal at Leavitt Partners and program manager at the CARIN Alliance, which is spearheading the efforts.”
CARIN is working with the Office of the National Coordinator for Health Information Technology and the Centers for Medicare and Medicaid Services (CMS), which will act as “government observers.”
From the FEHB front, benefits consultant Tammy Flanagan writes in Govexec about the Postal Service Health Benefits Program which will launch in 2025 as part of the Postal Reform Act of 2022. She observes
The version of the postal bill that eventually passed balances the risk pools, and the Office of Personnel Management now estimates premiums should go down for postal and non-postal employees and retirees alike.
The new law keeps all postal workers in FEHB, in their own group. All workers will be able to keep their current plans and avail themselves of the annual open season to choose other options within FEHB.
Future postal retirees will be required to enroll in Medicare A and B at 65. Retiree health coverage will then become a combination of Medicare and FEHB.
The question now is whether that requirement will eventually be extended to all federal employees, and what effect that would have on the premiums retirees pay. If that happens, at least federal employees will face one less tough decision at the time of retirement.
The FEHBlog expects that PSHBP premiums will be materially lower than FEHB premiums because PSHBP will accept Medicare funding for prescription drug benefits in the form of Part D EGWPs. Federal law has permitted the FEHB to offer premium-reducing Part D EGWPs for nearly twenty years. Nevertheless, OPM and a string of Administrations from George W. Bush to Joe Biden have refused to implement that law. Implementing that law in 2005 when it first took effect likely would have avoided the balkanization of the FEHB that we will soon experience with the PSHBP.
The FEHBlog does not expect the FEHB to adopt the mandatory Part B approach being taken by the PSHBP. Fewer retiring federal employees are picking up Part B because of the income-adjusted Part B premiums. As basic and income-adjusted Part B premiums continue to climb and climb, the FEHBlog expects that the PSHBP will liberalize, and then do away with, mandatory Part B. Meanwhile, the PSHBP’s undoubtedly favorable experience with Medicare funding of prescription drugs will lead OPM to allow FEHB the same opportunity.
With both branches of the Program using Part D EGWPs and integrated Medicare Advantage plans, everyone will enjoy reasonable premiums for high-quality healthcare. That in turn could lead to a reunion of the two branches. Hopefully, the PSHBP will be a relatively brief experiment that leaves the FEHB Program stronger.
The saving grace of the FEHB Program is that everyone in a plan option pays the same premium and the premiums are pooled to cover all plan option enrollees. That’s the bedrock principle of group health insurance that the FEHB Program has shown to work.