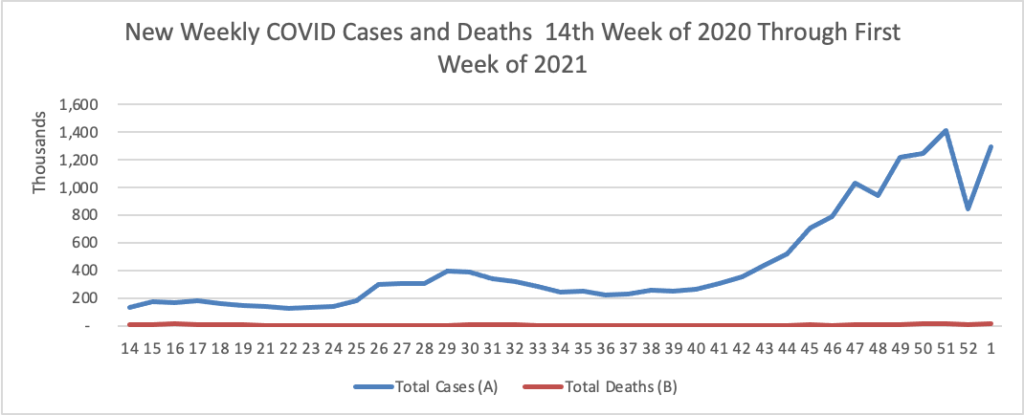
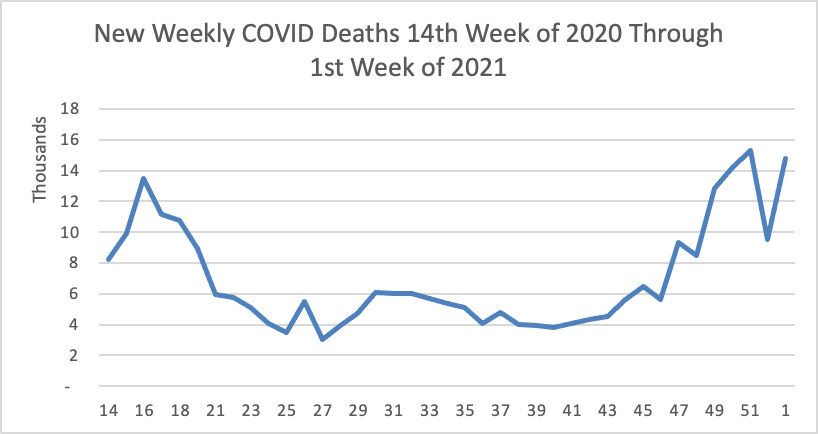
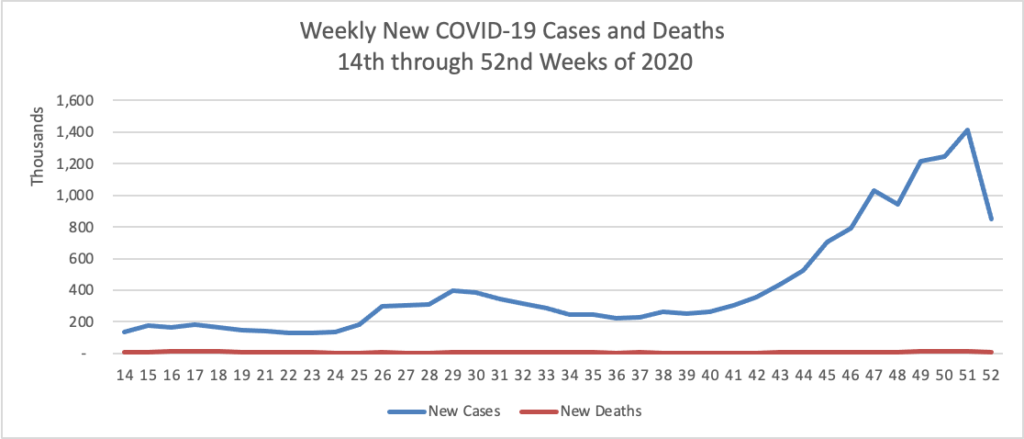
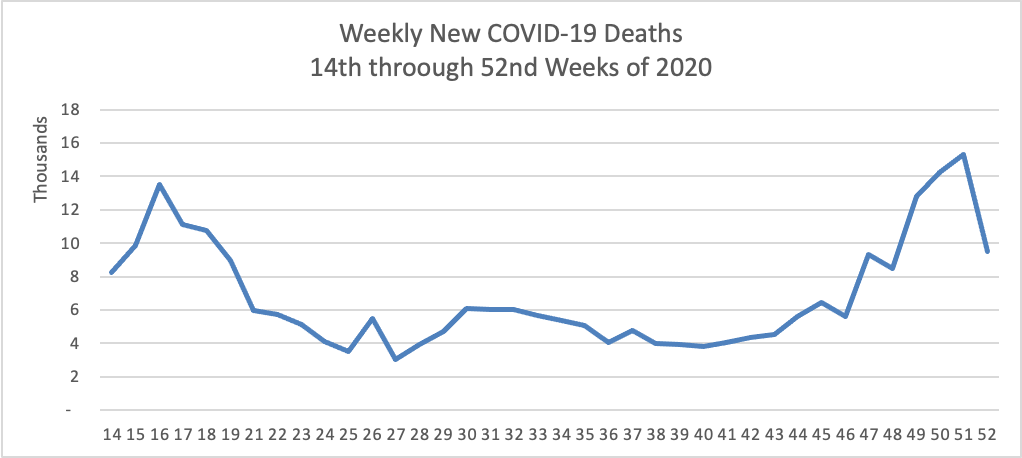
Tuesday Tidbits

Good news. The Wall Street Journal reports
The Trump administration is releasing second doses of coronavirus vaccines that were reserved for booster shots and is urging states to give the vaccine to anyone age 65 and older, as well as to people with pre-existing health conditions, a shift in strategy that vastly expands the pool of those eligible for shots while presenting another hurdle for the logistics of administering the vaccine at state and local levels.
Starting in two weeks, doses will be allocated based on the pace that states say they are administering the vaccine, as well as the size of a state’s population of people age 65 and older.
Health and Human Services Secretary Alex Azar announced the plan on ABC’s “Good Morning America” on Tuesday. “We now believe that our manufacturing is predictable enough that we can ensure second doses are available to people from ongoing production. So everything is now available to our states and our health-care providers,” he said.
The government had been allocating about half of newly available doses from Pfizer Inc. and Moderna Inc. to states since the vaccine rollout began last month. Federal officials said there is enough supply to ensure booster shots will be available and that they won’t extend the current recommended dosing schedules.
Drug makers expect to be able to produce enough vaccine doses to accommodate the new plans, vaccine manufacturers and supply-chain experts say.
The Journal also explains that while scientists are confident that the COVID-19 vaccines will protect you from contracting the disease, they don’t know yet whether they will prevent you from spreading the disease asymptomatically.
“Most vaccines prevent disease as opposed to preventing infection,” says Anna Durbin, a professor of international health at Johns Hopkins Bloomberg School of Public Health who is working on the AstraZeneca Covid-19 vaccine trial and previously worked on the Pfizer vaccine trial. She believes Covid vaccine studies will eventually show a reduction in asymptomatic transmission but not a complete elimination.
Even if vaccines don’t prevent transmission completely, they can still help populations achieve herd immunity if enough people take them, says Arnold Monto, an epidemiology professor at the University of Michigan School of Public Health who chairs the U.S. Food and Drug Administration’s Vaccines and Related Biological Products Advisory Committee. “We can still accomplish a lot even if it is demonstrated that there is still some asymptomatic infection occurring post-vaccination,” says Dr. Monto.
STAT News reports that the Pharmaceutical Care Management Association (“PCMA”) has asked the federal district court in DC to block a Trump Administration rule preventing the use of prescription drug manufacturer rebates in the Medicare Part D program. PCMA contends that the CMS rule impermissibly conflicts with a statutory exemption permitting the use of such rebates in Medicare Part D. Of course.
The Biden administration, which is set to take office on Jan. 20, could also choose not to defend the rebate policy in court, thus mooting the lawsuit.
Biden has not yet taken a position on eliminating drug rebates, but a number of prominent Democrats, including House Speaker Nancy Pelosi have spoken out against the policy as a ndout to the pharmaceutical industry.
Studies —
- Per the Wall Street Journal
The death rate from cancer in the U.S. dropped 2.4% from 2017 to 2018, the biggest single-year decline on record and a sign of the impact of new treatments on lung cancer especially, the American Cancer Society said.
It was the second year in a row with a record-setting drop, and the progress continues gains that have been made for more than a quarter-century, the cancer society said in a report published Tuesday. The researchers analyzed cancer mortality data from 1930 to 2018, before the start of the Covid-19 pandemic.
Overall, the cancer mortality rate has fallen 31% since its peak in 1991, according to the report, which was published online in the journal CA: A Cancer Journal for Clinicians. The latest decline left the mortality rate at 149 deaths for every 100,000 people in the general population in 2018, according to the American Cancer Society.
- Per the Rand Corporation
Use of telehealth jumped sharply during the first months of the coronavirus pandemic shutdown, with the approach being used more often for behavioral health services than for medical care, according to a new RAND Corporation study.
Between mid-March and early May 2020, telehealth was used by more than 40% of patients with a chronic physical health condition and by more than 50% of those with a behavioral health condition, according to findings published in the Journal of General Internal Medicine.
Overall, almost half of the people who were undergoing treatment when the pandemic shutdown began reported using some form of telemedicine.
Researchers found that the use of telehealth for behavioral health conditions was lower among women and among people over the age of 60. Use of telehealth also was lower among non-Hispanic whites relative to non-Hispanic Blacks, and was lower among those with less than a high school education relative to those with a college degree.