Thursday Miscellany

From Capitol Hill, the American Hospital Association gleefully reports that “The Senate today passed by 90-2 vote a bill that, among other health care provisions, would eliminate the 2% across-the-board cut to all Medicare payments, known as sequestration, until the end of 2021. To pay for the change, the bill, which was introduced by Sens. Jeanne Shaheen, D-N.H., and Susan Collins, R-Maine, would increase the fiscal year 2030 sequester cuts. The House is expected to take up the Senate-passed bill when it the week of April 13 when it returns to Washington D.C.”
The U.S. Office of Personnel Management announced six political appointments to the agency which do not require Senate confirmation. Good luck to them.
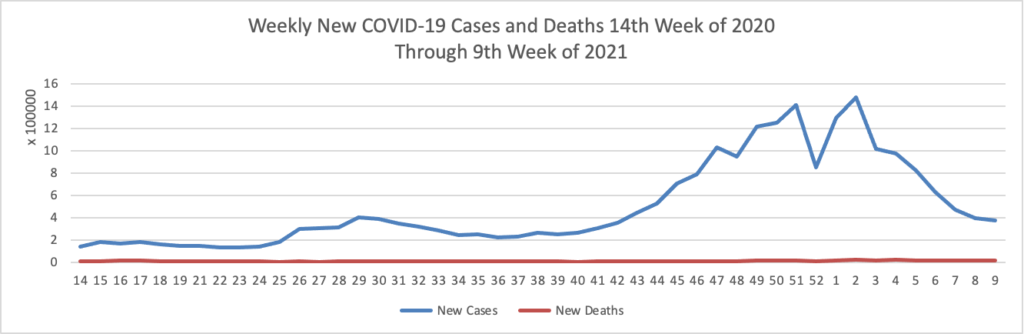
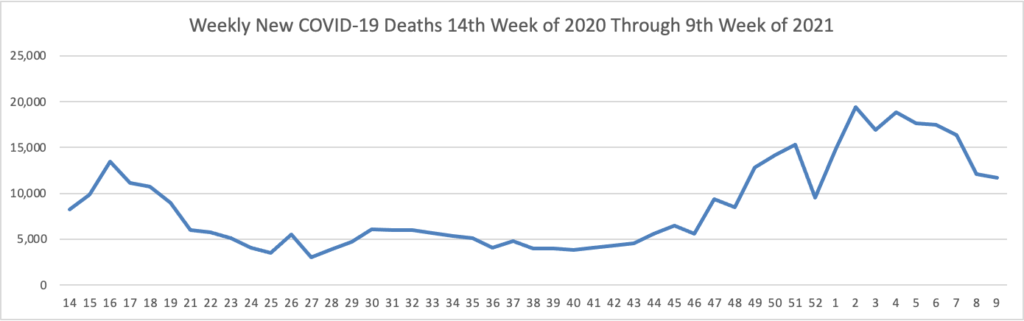
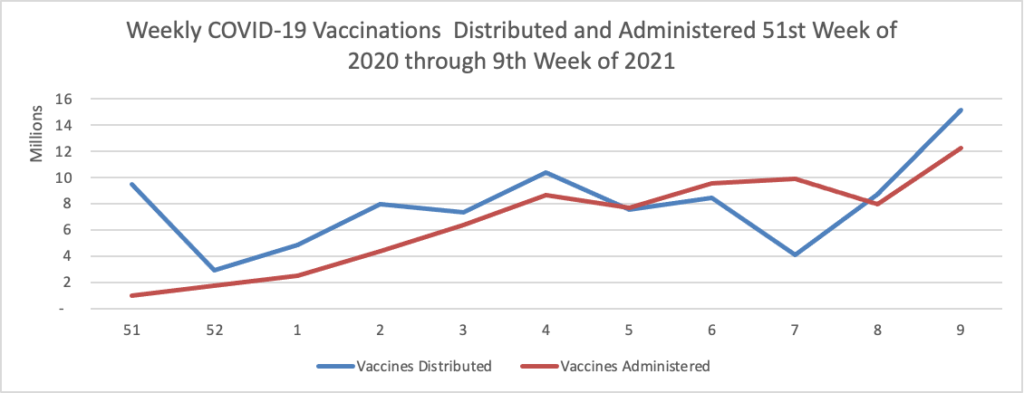
From the COVID-19 front, the Wall Street Journal observes
In many ways, AstraZeneca, which developed the vaccine in partnership with the University of Oxford, is delivering on its main promises. More than 70 countries, including the U.K. and much of the rest of Europe, have found the shot safe and effective. Although it isn’t a big player in vaccines, AstraZeneca helped make an experimental shot ready for mass use in less than a year. The company has built a manufacturing and distribution network that is delivering doses to the world’s poorest. Unlike most of its big competitors with vaccines or vaccine candidates, it has promised to do all this at no profit. The vaccine has been crucial to the U.K. drive that boasts one of the world’s best per-capita vaccination rates.
But at crucial moments, company executives have fumbled communications with governments, regulators and the public. That has left a reputational cloud over the vaccine effort—an effort that Dr. Soriot has said reflects the company’s desire to play a leading role in battling the pandemic.
The reverse — good public relations by a bad actor — would be a much worse situation. The FEHBlog hopes that the Food and Drug Administration does not delay emergency use authorization for the AstraZeneca vaccine.
Also from the COVID-19 front today, the Centers for Medicare and Medicaid Services issued a fact sheet on the value of monoclonal antibodies (mAb) treatment for high risk Covid-19 positive patients.
mAb treatment for COVID-19 is different from a COVID-19 vaccine. A vaccine triggers your body’s natural immune response, but can take weeks to develop enough antibodies and prevent some kinds of infection. Some vaccines for COVID-19 require two shots, so your body can develop its own immune response to the disease. But if you already have the virus, mAb treatment gives your body the antibodies it needs to protect itself.
That is positive news.
In miscellaneous healthcare news —
- Fierce Healthcare informs us that
Anthem is planning to acquire myNEXUS, a company that manages home-based nursing services for insurers.
According to the announcement, myNEXUS provides support to 1.7 million Medicare Advantage members across 20 states. The company’s platform largely automates the visit and authorization, getting care to the member faster, they said.
MyNEXUS uses a digital analytics tool in tandem with a team of more than 250 clinicians to plan and optimize home care, the companies said. In addition, it works with a nationwide network of providers and nursing agencies for local care.
- Healio reports that at “the Renal Physicians Association annual meeting, representatives from three companies [led by CVS Health] shared their approach to the changing paradigm of kidney care and emphasized the shift to value-based models that center on the patient.”
- America’s Health Insurance Plans announced that
Electronic prior authorization (ePA) can significantly reduce the time between a request for prior authorization and a decision and the time to a patient receiving care. These were two of the top findings from an initiative launched by America’s Health Insurance Plans (AHIP) to better understand the impact of ePA on the prior authorization process.
“Prior authorization is an important tool in helping patients receive safe, effective, clinically appropriate care,” said Kate Berry, Senior Vice President of Clinical Affairs at AHIP. “We are always looking for ways to enhance the patient and provider experience, and electronic prior authorization is an example. Today’s analysis provides a blueprint for how to leverage electronic tools to improve prior authorization.”
AHIP launched the Fast Prior Authorization Technology Highway—or Fast PATH—to better understand how electronic prior authorization could impact the process for patients and providers. Six health insurance providers—Blue Shield of California, Cambia Health Solutions, Cigna, Florida Blue, Humana, and WellCare (now Centene)—that collectively cover over 50 million Americans participated in the project, with Availity and Surescripts serving as the technology partners.
Bravo.
- Health Payer Intelligence informs us that “Payers should prepare for the payer price transparency rule to go into effect by building clear communication paths with members, reassessing their contracting processes, and asking themselves a couple of key questions, according to a report from PricewaterhouseCoopers’s Health Research Institute (HRI).” Check it out.