Weekend Update

From Capitol Hill, the House of Representatives and the Senate will be in session this week for Committee business and floor voting. Of note, the Senate will continue its voting process on the Postal Reform Act of 2022 (H.R. 3076) at 5:30 pm ET tomorrow. The continuing resolution funding the the federal government expires after 11:59 pm on Friday March 11.
The Wall Street Journal notes “House Democrats set to be in Philadelphia starting Wednesday for a long-planned retreat, negotiators need to make progress quickly.” The Journal’s article adds “Some aides and lawmakers of both parties said that the chances of passing an omnibus spending measure this week had increased, in part because a fiscal 2022 measure would include financing for Ukraine and Eastern European allies and give the military flexibility for any related needs.”
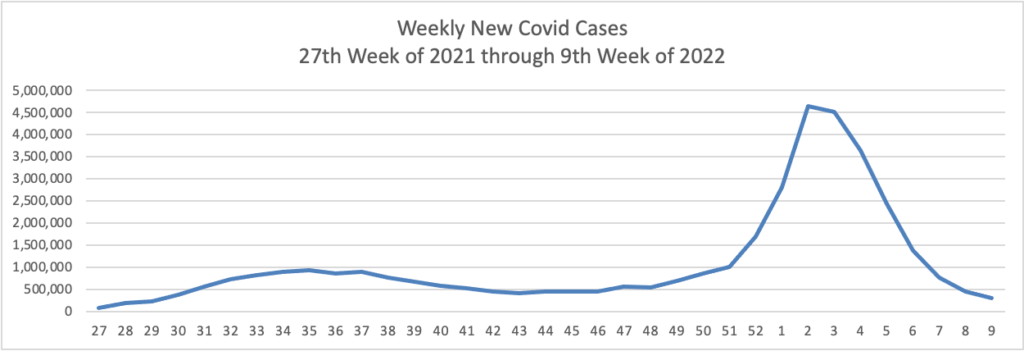
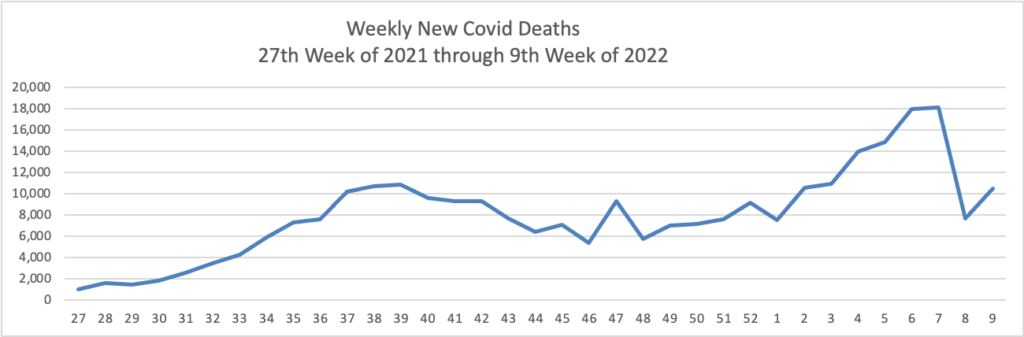
From the Omicron front —
- The New York Times reports on the difficulties encountered in the distributing the Evusheld antibiotic treatment that shields immunocompromised Americans who cannot receive a Covid vaccination. According to the article, immunocompromised Americans represent 3% of the population.
- The Wall Street Journal informs us
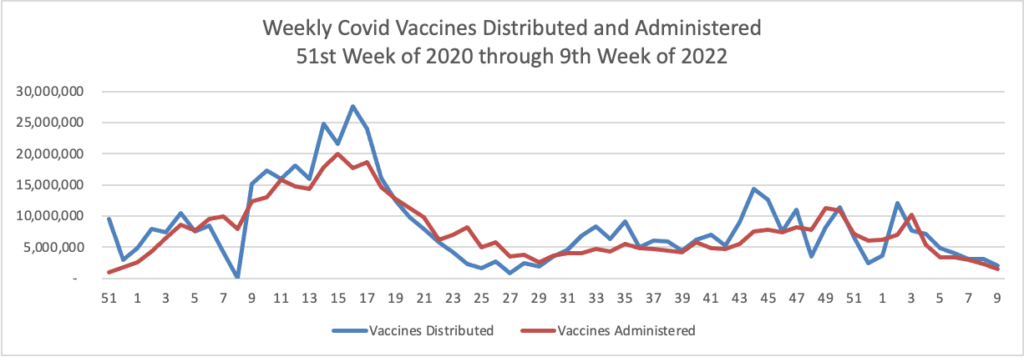
Novavax Inc.’s long-awaited Covid-19 vaccine is moving toward U.S. authorization after the company said it resolved manufacturing problems that had held up its application.
Clearance in the U.S. isn’t imminent because the Food and Drug Administration must sort through a large amount of study data from several countries, a person familiar with the matter said. A decision is getting closer, however, since Novavax formally submitted an authorization request in late January.
If the shot is rolled out in the U.S., it could boost vaccination efforts that have flagged among the hesitant, according to Novavax and doctors.
- The Journal also tells us
Vaccination campaigns have lagged behind in much of the developing world, as many countries struggle to distribute shots outside major urban areas. In Asia, countries like India, Indonesia and the Philippines have fully vaccinated half their populations. That is higher than the 13% of people living in Africa who are vaccinated, with 4% immunized in Nigeria, the continent’s most populous country. Latin America has done better. Many countries, including Brazil and Peru, have vaccinated more than 70% of their people.
Natural immunity, which refers to antibodies acquired through infection, was widespread in Indonesia when Omicron arrived. One study from October to December of roughly 20,000 Indonesians found that 74% of unvaccinated Indonesians had protective antibodies, according to Pandu Riono, a University of Indonesia epidemiologist who worked with government researchers on the study.
Based in part on the results, Mr. Riono and his colleagues estimate that 70% of Indonesians had been infected with Covid-19 as of December, far higher than the 34% of Americans who had been infected by then, according to a survey by the U.S. Centers for Disease Control and Prevention.
From the federal employment front, Federal News Network offers a transcript of an interview between Federal News Network reporter Tom Temin and American University professor Bob Tobias about the demographic characteristics of federal employees. For example, “74% of the workforce, federal workforce is 40 or older, versus 54% in the private sector.” Unquestionably, the experience of the federal employee population benefits all of us, but older workforce also impacts FEHB premiums.
From the telehealth front, Fierce Healthcare reports
Striking the balance between virtual and in-person care is a key trend to watch heading into the rest of 2022, according to a new survey from Evernorth.
Ipsos surveyed 3,000 consumers, 575 human resources professionals and 58 health plan leaders on behalf on the Cigna subsidiary. The report underscores patients’ growing comfort with virtual care and telemedicine in the wake of COVID-19, with 75% saying they agree more care will be provided virtually; 57% of those surveyed said they used virtual care over the last year.
However, consumers are not enthusiastic about virtual options in all settings, according to the survey. While 35% of those surveyed said they were satisfied with their experience in virtual primary care, just 11% said the same about behavioral health care. Nineteen percent said they were satisfied with virtual care for specialty services, while 5% said the same for physical therapy and 6% for nutrition and diet services.
While satisfaction with virtual behavioral health care was mixed in the survey, a growing number of people are seeking behavioral health services in virtual settings.
Boom!
From the strategery front, Becker’s ASC News discusses the 2022 growth strategies of Optum’s three business divisions:
- Optum Health, the healthcare provider division which includes physician groups and ambulatory surgery centers
- Optum Insight, which houses the data analytics platforms designed to connect clinical, administrative and financial data, and
- Optum Rx, a pharmacy benefits and care services business.