Thursday Miscellany

From Capitol Hill
Roll Call tells us
Senators negotiating a COVID-19 supplemental funding package have an “agreement in principle” to provide roughly $10 billion for the Department of Health and Human Services to stock up on waning domestic supplies for combating the virus, according to Sen. Roy Blunt, R-Mo.
Blunt, the top Republican on the Senate Labor-HHS-Education Appropriations Subcommittee, said both parties have tentatively agreed to offsets for the $10 billion that would repurpose unspent funds from prior pandemic relief laws.
The offsets negotiators agreed to include $2.2 billion from unused grant funds for venues like zoos and theaters and $2 billion in untapped assistance to the aviation and manufacturing industry, Blunt said. His comments to reporters came after a Republican Conference lunch in which lead GOP negotiator Sen. Mitt Romney of Utah briefed his colleagues on the agreement in principle.
Fierce Healthcare informs us
The House [of Representatives] passed a bill on Thursday that caps the out-of-pocket cost of insulin at $35 a month for beneficiaries in Medicare Part D and for certain group and individual plans.
The Affordable Insulin Now Act, which passed the House via a 232 to 193 vote, comes as work in the Senate continues on a bipartisan alternative that could bring additional changes. * * *
Private plans would also be required to offer first-dollar coverage of insulin without any deductible, according to an analysis from the Congressional Budget Office.
The cap on cost-sharing for private insurance plans would implement in 2023.
The Hill offers a related article explaining why insulin prices are so “troubling” high.
HR Dive reports
Among the provisions of this month’s $1.5 trillion omnibus spending bill, Congress included a revival of an exemption that allowed high-deductible health plans to cover telehealth before individuals meet their deductible.
The provision was originally created by the Coronavirus Aid, Relief, and Economic Security Act, which sunset at the end of 2021. The provision will resume April 1 but will again sunset at the end of this year.
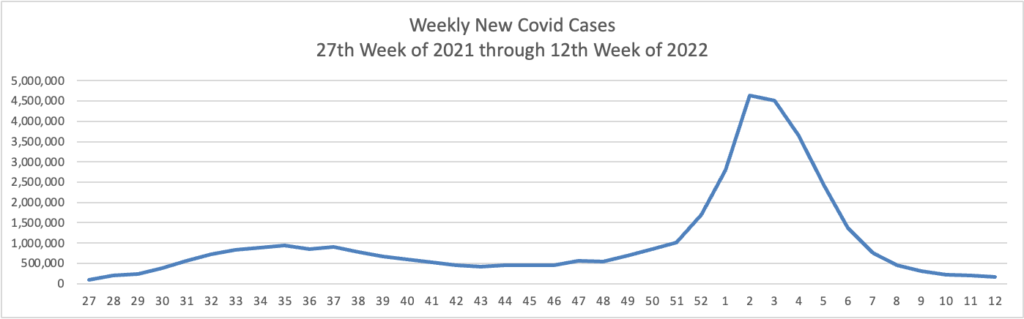
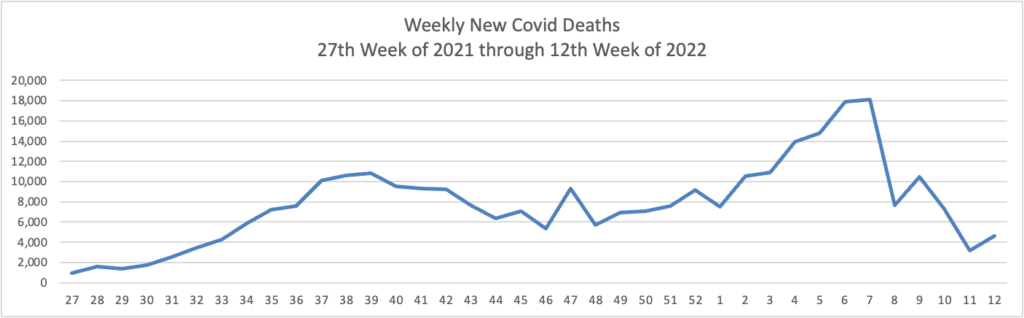
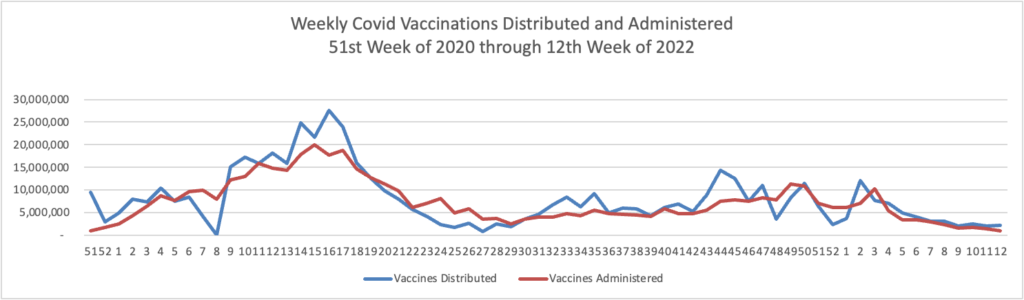
From the Omicron and siblings front, the Wall Street Journal discusses the state of Covid treatments and offers its advice on who should seek out a second Covid booster besides the immunocompromised.
From the OPM front, Govexec projects OPM’s actions over the next 18 months based on the FY 2022 to FY 2026 strategic plan released last Monday. In short, “Hire, Hire, Hire.”
Govexec also discusses efforts underway by OPM, the Social Security Administration and the Thrift Savings Plan to improve the customer service experience of federal employees and retirees. Good luck with that.
From the research front the National Institutes of Health announced
Scientists have published the first complete, gapless sequence of a human genome, two decades after the Human Genome Project produced the first draft human genome sequence. According to researchers, having a complete, gap-free sequence of the roughly 3 billion bases (or “letters”) in our DNA is critical for understanding the full spectrum of human genomic variation and for understanding the genetic contributions to certain diseases. The work was done by the Telomere to Telomere (T2T) consortium, which included leadership from researchers at the National Human Genome Research Institute (NHGRI), part of the National Institutes of Health; University of California, Santa Cruz; and University of Washington, Seattle. NHGRI was the primary funder of the study.
Analyses of the complete genome sequence will significantly add to our knowledge of chromosomes, including more accurate maps for five chromosome arms, which opens new lines of research. This helps answer basic biology questions about how chromosomes properly segregate and divide. The T2T consortium used the now-complete genome sequence as a reference to discover more than 2 million additional variants in the human genome. These studies provide more accurate information about the genomic variants within 622 medically relevant genes. * * *
The now-complete human genome sequence will be particularly valuable for studies that aim to establish comprehensive views of human genomic variation, or how people’s DNA differs. Such insights are vital for understanding the genetic contributions to certain diseases and for using genome sequence as a routine part of clinical care in the future. Many research groups have already started using a pre-release version of the complete human genome sequence for their research.
From the mental healthcare front, the American Hospital Association calls our attention to a new GAO report.
Consumers with health coverage experience challenges finding in-network mental health providers, who may not be accepting new patients or have long wait times to see them, according to a new report from the Government Accountability Office. Factors contributing to these challenges include low reimbursement rates for mental health services and inaccurate or out-of-date information on provider networks, GAO said. The report also looks at ongoing and planned federal efforts to address these challenges, for example by increasing the mental health workforce, mental health system capacity and oversight of health plan compliance with mental health parity laws.
This squib caused the FEHBlog to recall a comment that he heard at a conference — Four out of five doctors are in-network but only one out of five mental health providers are in-network with the notable exception of hub and spoke telemental services.
Yesterday the FEHBlog suggested that in return for three free primary care visits and three behavioral health visits, plan members should name and use their in-network primary care provider and primary mental health provider. The FEHBlog is sticking with this idea for the in-network primary care provider but he recognizes the idea may be premature for the in-network primary mental health provider. Of course, creating a looser standard for free mental health care compared to primary care is compliant with the federal health parity rule. The reverse would violate the often fuzzy non-quantitative treatment limitations created by the law.
From the miscellany department —
- STAT News tells us “With Medicare expected to cover a projected 80 million people by 2030,He entrepreneurs and investors are cashing in on what analysts see as an inevitable shift in health care away from the hospital and into the homes of aging patients.” The publication identifies five related technology trends.
- Fierce Healthcare reports “Virtual care startup Hims & Hers is teaming up with Carbon Health to offer patients in California with direct access to providers for in-person medical appointments at clinics. The collaboration will provide easy and comprehensive access to a broader range of care options through the Hims & Hers platform, company executives said.”