Thursday Miscellany

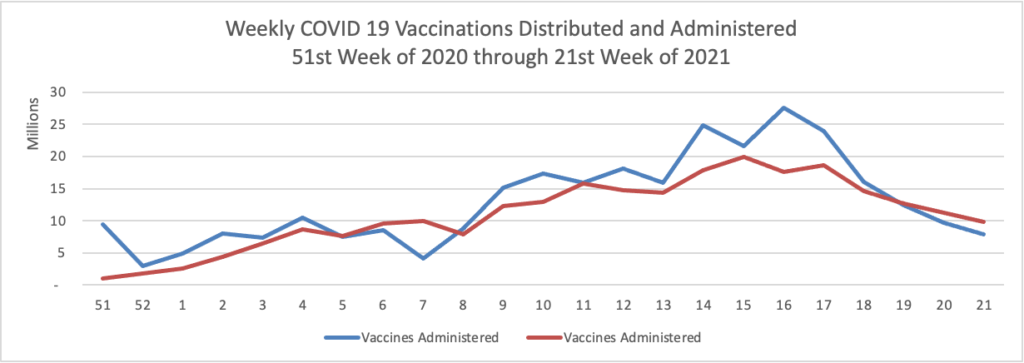
President Biden today announced a strategy for sharing “at least 80 million U.S. [COVID 19] vaccine doses globally by the end of June.
On the U.S. COVID-19 vaccination front
- David Leonhardt reports in the New York Times that
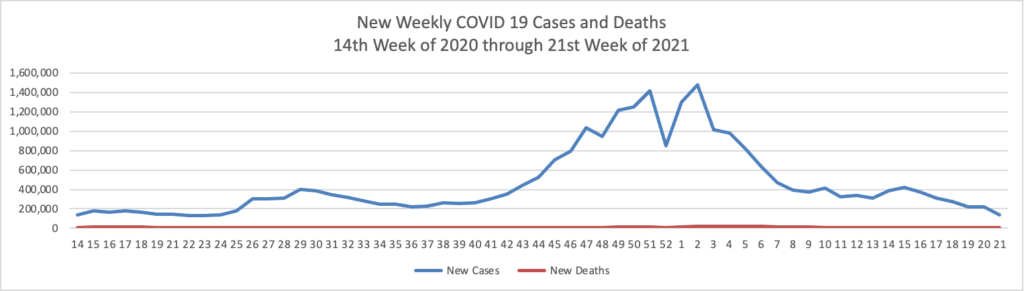
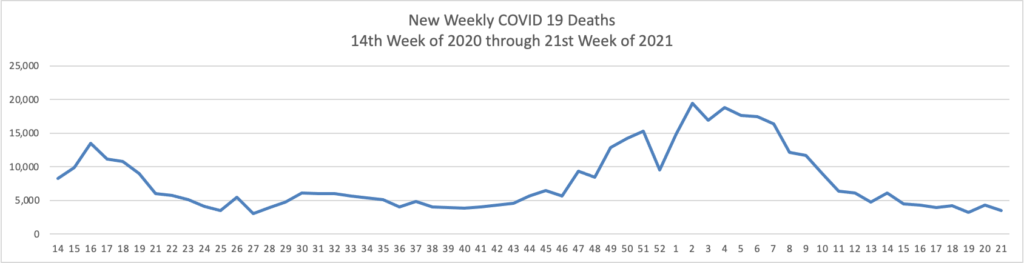
When the C.D.C. reversed its Covid-19 guidelines last month and said that vaccinated Americans rarely needed to wear masks, it caused both anxiety and uncertainty.
Many people worried that the change would cause unvaccinated people to shed their masks and create a surge of new cases. On the flip side, a more optimistic outcome also seemed possible: that the potential to live mostly mask-free would inspire some vaccine-hesitant Americans to get their shots.
Almost three weeks after the change, we can begin to get some answers by looking at the data. So far, it suggests that the optimists were better prognosticators than the pessimists.
- HR Dive informs us that “Vaccine mandates are not a consideration for 83% of employers responding to law firm Fisher Phillips’ recent pulse survey. That figure is up from January, when the firm recorded 64% of respondents saying they would not impose a vaccine requirement. At that time, 27% of employers said they had yet to decide if they would mandate vaccinations. “Most employers — 75% — said they are encouraging workers to get their vaccines, the May 25 survey results revealed.”
Fortune Magazine released its Fortune 500 and sitting in the top 10 are four healthcare companies (CVS Health (4), United Health Group (5), McKesson (7) and AmerisourceBergen (8). For the second year in a row the top two companies are Walmart and Amazon, both of which are attempting to break into the healthcare market.
In his latest FedWeek column, Reg Jones discusses continuing FEHB coverage of unmarried children of enrollees beyond age 26 provided the child is incapable of self-support. “The term “incapable of self support” generally means that the child earns less than the equivalent of GS-5, step 1 [$30,414 in 2021]. However, this is not a hard and fast rule. In making a decision, consideration is given to the child’s earnings and condition or prognosis.”
From the Centers for Medicare and Medicaid Services front:
- Kaiser Health News reports that expanding health insurance coverage is the top priority of the newly installed CMS Administrator, Chiquita Brooks-Lasure.
- Healthcare Dive tells us that Elizabeth Fowler, head of the Center for Medicare and Medicaid Innovation, said that CMMI’s ongoing strategy review has resulted in more conscious choices in where it should invest, which includes pivoting away from voluntary models. “Voluntary models are subject to risk selection, which has a negative impact on the ability to generate system-level savings. Providers that aren’t generating the extra revenue tend to exit the program, and those that are tend to stay,” Fowler, now on her third month at the job, said at a Health Affairs briefing. “So we are exploring more mandatory models.”
- Fierce Healthcare informs us that Congress wants CMMI to be more transparent in its spending. A bipartisan letter “said CMMI’s authorizing statute, which was part of the Affordable Care Act, calls for the center to gather input from interested parties. However, this requirement has often been shunted aside by the center and rarely observed.”
Medcity News reports that the Labor Department’s top health benefits law enforcement priority is compliance with the federal mental health parity law.
To uphold the law and ensure parity in coverage, the labor department has two strategies in place, [Secretary of Labor] Walsh said.
The department’s Employee Benefits Security Administration agency has created a task force that focuses on enforcement of the act, he said. The task force is reviewing its inventory of case files, looking to identify potential violations and send out requests to payers for data on parity analyses, which they are required to maintain to show their compliance with the law.
Further, the Department of Labor, along with other government agencies involved in this work such as the Department of Health and Human Services and Internal Revenue Service, is providing regular reports to Congress on their findings and enforcement actions, Walsh said. This can help inform legislation on insurance coverage moving forward.
Govexec provides the latest Postal Service news, including a confirmed report that the FBI is investigating Postmaster General DeJoy “for allegations that he illegally pressured employees at his former company to donate to Republican candidates while promising to later reimburse them through bonuses.”
The Wall Street Journal reports today that
A drug sold by AstraZeneca PLC and Merck & Co. reduced the recurrence of breast cancer in women with an early but aggressive form of the disease, a long-running [blinded] international study found. The finding, which on Thursday was published online by the New England Journal of Medicine and released at a major cancer-research meeting, marked the latest advance in cancer treatments targeting the genetic traits of tumors. It could expand the arsenal of weapons against a hereditary form of breast cancer. The result also helps validate the pharmaceutical industry’s investment in a pricey new class of drugs that target cancer cells, known as PARP inhibitors. * * *
PARP inhibitors work by blocking cancer cells from relying on a survival tactic: the ability to repair their own DNA after their DNA is damaged naturally or by other drug treatments. This, in turn, contributes to cancer-cell death.
Health regulators have approved these types of drugs in recent years to treat ovarian, breast, prostate and pancreatic cancers. The drugs have been found to be particularly useful against cancers associated with harmful mutations in genes known as BRCA1 and BRCA2. Women with these hereditary mutations have a higher risk of developing breast cancer, and often at a younger age than is typical. The BRCA mutations account for about 5% of the estimated 281,000 cases of breast cancer diagnosed annually in the U.S.