Tuesday’s Tidbits

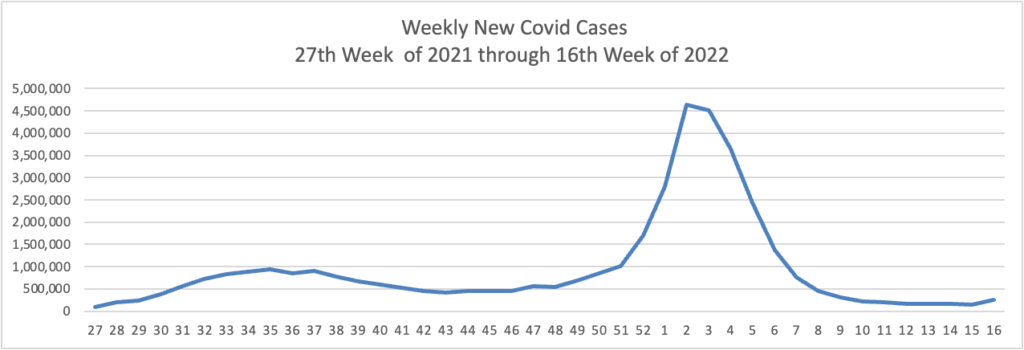
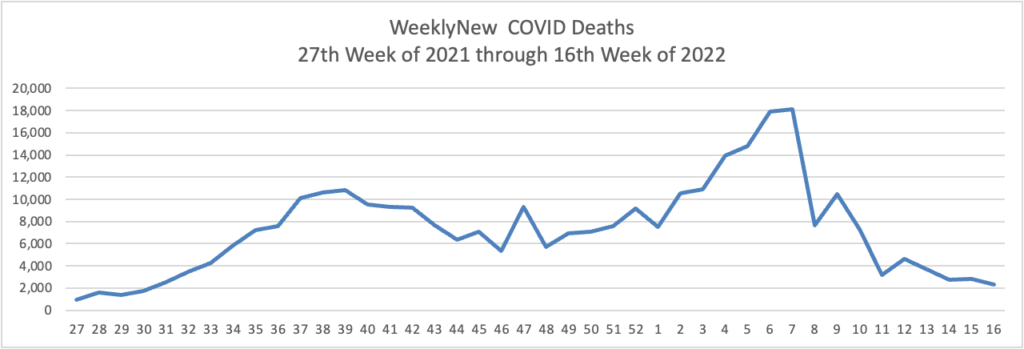
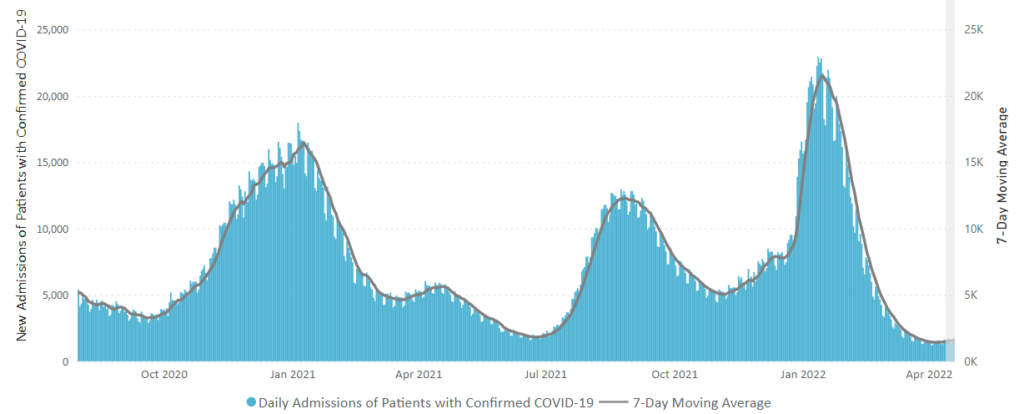
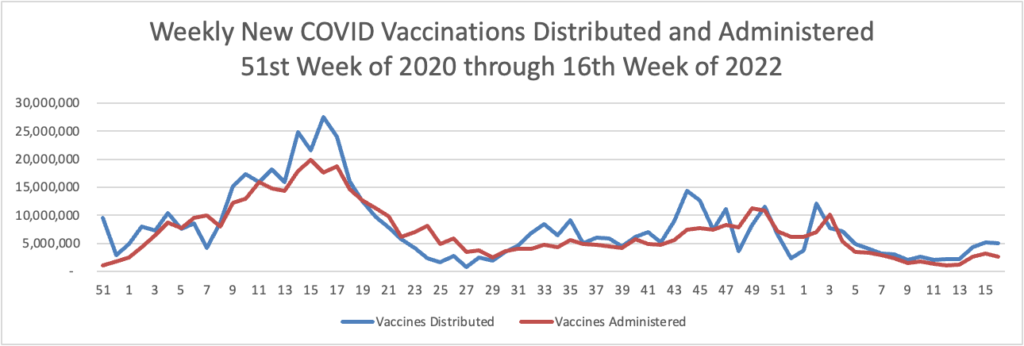
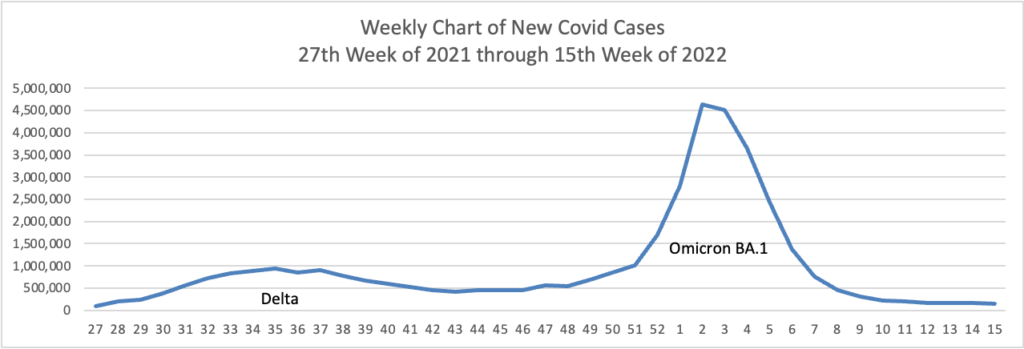
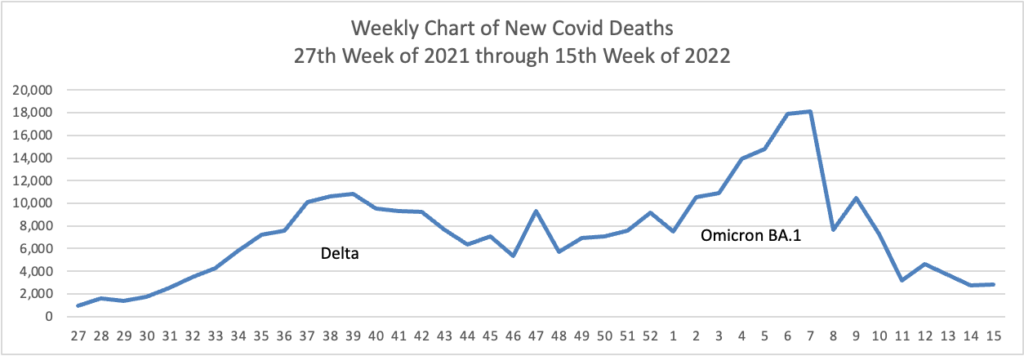
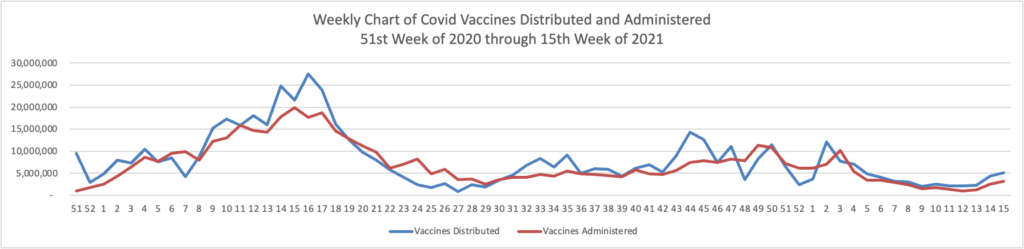
From the Omicron and siblings front —
The American Hospital Association informs us
The share of the U.S. population with antibodies to the SARS-CoV-2 virus in their bloodstream increased from 34% in December 2021 to 58% in February 2022, including 75% of children, according to a study released today by the Centers for Disease Control and Prevention. The study tested blood samples during the COVID-19 omicron period for antibodies produced in response to infection but not in response to COVID-19 vaccines. Children had the highest rates of infection-induced antibodies and adults 65 and older the lowest, with the greatest increases over the period in age groups with the lowest COVID-19 vaccination coverage.
“Vaccination remains the safest strategy for preventing complications from SARS-CoV-2 infection, including hospitalization among children and adults,” the authors said.
and
Starting this week, the Administration will allow all pharmacies in the federal pharmacy program to order free oral antiviral treatments directly from the federal government, the White House announced today. The Administration hopes to double the number of participating pharmacies to 40,000 in the coming weeks, and to launch new Test-to-Treat locations that offer the Pfizer and Merck pills, which the Food and Drug Administration authorized in December to treat COVID-19 in patients at risk of progressing to severe disease. Pharmacies also can continue to receive the pills through their state or territorial health department. The Administration said it is working to improve the Test-to-Treat patient experience, including through telehealth options; and to provide more guidance on COVID-19 treatments to prescribers and clinicians.
The New York Times adds more details to this AHA blurb. In short, “experts say that efforts to reach at-risk Americans remain complex and inefficient.”
Medscape adds “Contrary to popular belief, no association appeared between the number of intensive care unit beds and COVID-19 deaths, based on a review of data from all 50 states between March 1, 2020, and June 30, 2021.”
The Wall Street Journal further reports
Pfizer Inc. and partner BioNTech SE asked U.S. health regulators to authorize a booster dose of its Covid-19 vaccine for children 5 to 11 years old.
The request Tuesday to the Food and Drug Administration comes after the companies said earlier this month that a third shot safely generated a strong immune response in the youngsters, including significantly increased antibody levels against the Omicron variant.
A thumbs-up from the FDA would expand eligibility of boosters to the roughly 28 million children in the U.S. Booster doses are now available for people as young as 12 years old in the U.S., and regulators recently greenlighted second boosters for people who are 50 years old and up or who have weakened immune systems.
In FEHB news, OPM issued a paper describing the 2021 highlights of OPM’s FEHB Plan Performance Assessment system. In the FEHBlog’s view, OPM’s PPA system could be improved by (1) seeking plan input on all PPA changes, including, for example, the benchmark change to ALOB and (2) implementing changes for the first measurement year following the change, not the year in which the change is made. Both of these changes are consistent with federal administrative law, in the FEHBlog’s opinion. Also, OPM should use the carrot incentive more than the stick.
In healthcare business news,
Healthcare Dive tells us
Hospitals are experiencing a “massive surge” in expenses for items such as labor, drugs and supplies amid rising inflation, the American Hospital Association said in a report on Monday.
Labor is a particular stressor, making up more than half of hospitals’ total expenses. Overall, hospital labor expenses per patient increased almost 20% from 2019 to 2021, the AHA said.
The powerful hospital lobby urged Congress to help address these headwinds by adding money to the provider relief fund and creating flexibility on advanced Medicare repayments, among other items.
Medpage Today informs us
Nearly three-fourths of U.S. physicians opted for employment with hospitals, health systems, or other corporate entities, such as private equity firms and health insurers in the pandemic era, according to a new report.
In 2021, 73.9% of physicians were hospital- or corporate-employed, up from 69.3% at the start of 2021, 64.5% at the start of 2020, and 62.2% at the start of 2019, according to the nonprofit Physicians Advocacy Institute (PAI) and consulting firm Avalere. That equates to 484,100 employed physicians, up from 423,800, 391,000, and 375,400 at the start of 2021, 2020, and 2019, respectively.
Perhaps these two trends are related? On the one hand, more physician employees create more hospital expenses. On the other, a hospital receives additional health plan payments for services provided by physician employees. In all likelihood, the revenue exceeds the expense in this case.
From tidbits department,
- AHRQ reports on “Geographic Variation in Inpatient Stays for Five Leading Mental Disorders, 2016–2018.”
- EHR Intelligence reports
CAQH CORE, the author of national operating rules for the HIPAA-covered administrative transactions, recently released new operating rules to enhance information exchange and healthcare operations related to benefits coverage and supplemental documentation. * * *
The new CAQH CORE Attachments Operating Rule aims to improve the exchange of attachments, a long-standing industry issue. The guidelines will establish key infrastructure and data content requirements, helping providers send electronic health plans documentation to support a claim or prior authorization in a uniform format, the press release stated.
Reassociation or linking the attachment with the original prior authorization request or claim submission is one of the most significant pain points in the attachment workflow, CAQH CORE added.
The new guidelines also offer updates to enhance the exchange of critical eligibility and benefit information related to telemedicine, prior authorization, remaining coverage benefits, procedure-level information, and tiered benefits between health plans and providers.
The second newly released rule, the CAQH CORE Eligibility & Benefits Data Content Rule, intends to enhance provider knowledge regarding their patients’ coverage, leading to more timely care and accurate billing.
Finally, CAQH CORE revised its rules for infrastructure, which now calls for greater health plan system availability and less frequent periods of downtime.