Friday Report

Happy Washington’s Birthday (one day early)
First in war, first in peace, first in the hearts of his country
From Washington, DC,
- “The American Medical Association News reports,
- “The U.S. Senate voted 52-48, after a 10-hour “vote-a-rama” session, to adopt a budget resolution for fiscal year 2025 focusing on the border, military and energy. The bill would authorize roughly $340 billion in spending and be fully offset by corresponding spending cuts. The budget resolution is a blueprint for one of two budget reconciliation bills the Senate hopes to enact this year, with the second focusing on extending tax cuts and cutting spending.
- “Meanwhile, the House of Representatives next week plans to vote on its own budget resolution focusing on the Trump administration’s agenda on border security, defense, energy and taxes. The budget resolution calls for $2 trillion in spending cuts that could potentially impact Medicaid and other key health care programs. The proposal also allows for up to $4.5 trillion in spending for tax cuts. President Trump this week expressed his preference for the House’s one-bill approach.” * * *
- “Both chambers must pass a common budget resolution to move forward with the reconciliation process.”
- MSN lets us know,
- “President Donald Trump warned drugmakers in a private meeting [yesterday] that tariffs are coming and said companies should hustle to move overseas manufacturing to the US, according to two people familiar with the conversation.
- “Trump also didn’t commit to pushing Congress to water down a drug pricing program enacted under President Joe Biden that the pharmaceutical industry has been seeking relief from.
- “The president’s tone suggests the pharmaceutical industry’s bid to win an ally in the White House might be more difficult than executives had hoped. Despite his pro-business leanings, Trump had a rocky relationship with drug companies in his first term, at one point accusing them of “getting away with murder” on the price of medicines.”
- The Wall Street Journal informs us,
- The U.S. Food and Drug Administration declared that the shortage of Novo Nordisk’s weight-loss medications Wegovy and Ozempic has been resolved, according to its website.
- The FDA announcement confirms that the U.S. supply of both prescription-only drugs now meets or exceeds the current and projected demand in the country, Danish pharmaceutical company Novo Nordisk said Friday.
- The popularity of Novo Nordisk’s blockbuster GLP-1 drugs had forced the FDA to include Wegovy and Ozempic in its official shortage list, despite the company’s efforts to ramp up production.
- Ozempic, which treats diabetes, and Wegovy, sold for obesity, have spent over two years on the FDA’s list. During that time, compounding pharmacies–those which produce custom medications–were legally entitled to produce copies of both semaglutide drugs, which were also cheaper than Novo Nordisk’s versions.
- The regulator’s decision to take them off of the shortage list means that compounding pharmacies will need to stop making and selling knock offs.
- Modern Healthcare adds
- “The Food and Drug Administration issued a notice on Friday classifying its recall of the Boston Scientific Accolade pacemaker devices as the most serious type of recall.
- “To date, 832 injuries and two deaths tied to the devices have been reported. The recall affects about 13% of Accolade devices manufactured before September 2018.
- “The pacemakers treat slow heart rhythms by pacing the upper and lower chambers of the heart and adjusting the pacing rate to meet the body’s needs. Due to a manufacturing issue with the battery cathode, the pacemakers might enter safety mode under certain conditions, and as a result may not properly regulate the heart’s rhythm and rate, according to the FDA.”
- Govexec tells us,
- “The Office of Personnel Management is laying off its entire procurement team as the federal government’s human resources agency continues to reduce its footprint.
- “Employees were informed Friday that their positions were being “abolished” and they would be separated from federal service in 60 days. The decision is separate from the ostensibly for-cause firings that OPM kicked off internally earlier this month and have subsequently swept up agencies throughout the government.
- “The employees impacted by Friday’s moves received reduction-in-force notices from acting OPM Director Charles Ezell, who said he was issuing the layoffs due to three separate executive orders issued by President Trump.”
- The IRS issued guidance about “Health Coverage Reporting Required by Sections 6055 and 6056,” the IRS 1095-B and 1095-C forms following up on a statutory change that occurred last December.
From the judicial front,
- Politico reports.
- “A federal judge blocked President Donald Trump’s bid to deprive federal funding from programs that incorporate “diversity, equity and inclusion” initiatives.
- “U.S. District Judge Adam Abelson ruled that Trump’s policy likely violates the First Amendment because it penalizes private organizations based on their viewpoints. And the judge said the policy is written so vaguely that it chills the free speech of federal contractors concerned they will be punished if they don’t eliminate programs meant to encourage a diverse workforce.
- Per Govexec,
- “A federal judge in Washington, D.C., on Friday ended a weekslong-halt on the Trump administration’s plan to put the vast majority of employees at the U.S. Agency for International Development on administrative leave, denying unions’ request to issue a preliminary injunction in the case.
- “The American Foreign Service Association and the American Federation of Government Employees sued to block the apparent effort to decimate the agency and reposition it under the auspices of the State Department. More than 2,000 employees were briefly placed on paid administrative leave before the court’s initial intervention earlier this month, and another 2,000 workers mostly stationed overseas also are on the at-least-temporary chopping block.
- “U.S. District Judge Carl Nichols, a Trump appointee, had devoted most of his attention during hearings to concerns regarding the continued safety of those overseas workers stationed in high-risk regions. But recent filings from Peter Marocco, the agency’s day-to-day chief under Acting Administrator and Secretary of State Marco Rubio, stating that overseas employees will continue to have access to security-related systems like the SAFE Alert system and the SCRY Panic smartphone app, assuaged the judge’s fears.”
- Beckers Hospital Review relates,
- “The Trump administration plans to defend the ACA requirement that requires insurers to fully cover certain preventive services for their members.
- “On Feb. 18, the Justice Department filed a brief with the Supreme Court, arguing in favor of maintaining the Biden administration’s stance regarding the landmark case that centers on whether employers can exclude covered services on religious grounds. Specifically, the case addresses the authority of the U.S. Preventive Services Task Force (USPSTF) in mandating coverage for preventive services, including medications such as PrEP for HIV prevention.”
From the public health and medical research front,
- The Centers for Disease Control and Prevention announced today,
- “Seasonal influenza activity remains elevated across the country. COVID-19 activity is elevated in many areas of the country. RSV activity is declining in most areas of the country.
- “COVID-19
- “COVID-19 activity is elevated in many areas of the country. Though wastewater levels are high, emergency department visits are at low levels, and laboratory percent positivity is stable. Emergency department visits and hospitalizations are highest in older adults and emergency department visits are also elevated in young children.
- “There is still time to benefit from getting your recommended immunizations to reduce your risk of illness this season, especially severe illness and hospitalization.
- “CDC expects the 2024-2025 COVID-19 vaccine to work well for currently circulating variants. There are many effective tools to prevent spreading COVID-19 or becoming seriously ill.
- “Influenza
- “Seasonal influenza activity remains elevated across the country. Additional information about current influenza activity can be found at: Weekly U.S. Influenza Surveillance Report | CDC.
- “RSV
- “RSV activity remains elevated but is declining in most areas of the country. Emergency department visits and hospitalizations are highest in children and hospitalizations are elevated among older adults in some areas.
- “Vaccination
- “Vaccination coverage with influenza and COVID-19 vaccines is low among U.S. adults and children. Vaccination coverage with RSV vaccines remains low among U.S. adults. Many children and adults lack protection from respiratory virus infections provided by vaccines.
- “Additional Respiratory Illnesses
- “Pertussis
- “Reported cases of whooping cough (pertussis) continue to be elevated nationwide. Whooping cough is very contagious and can spread easily from person to person. Babies younger than 1 year old are at highest risk of severe disease and complications. The best way to prevent complications from whooping cough is to get vaccinated. Learn more: About Whooping Cough | Whooping Cough | CDC.
- “Mycoplasma pneumoniae
- “Respiratory infections caused by the bacteria Mycoplasma pneumoniae have declined from their peak in late 2024 but remain high nationwide, especially in young children. M. pneumoniae infections are generally mild but can sometimes be severe, causing what’s known as “walking pneumonia.” Most people will recover without medicine, but some need antibiotics to get better. Learn more: About Mycoplasma pneumoniae Infection | M. pneumoniae | CDC.
- “Group A Strep
- “Respiratory infections caused by group A Streptococcus bacterium, such as strep throat and scarlet fever, are elevated nationwide. This is typical for this time of year. Healthcare providers can do a quick test to see if someone has strep throat or scarlet fever and if treatment with antibiotics can help. Learn more: About Strep Throat | Group A Strep | CDC.
- “Pertussis
- Season Outlook
- “The CDC has determined that the highest hospital demand for COVID-19, flu, and RSV to date this season occurred during the week ending February 1, 2025, and it was lower than the peak demand from last season. However, influenza activity remains high in most areas.
- “The peak hospital demand due to COVID-19 during this fall and winter respiratory season was lower than all previous seasons, and nearly 50% lower than the peak demand last season.
- “CDC does not anticipate producing additional respiratory disease outlook updates during the remainder of the 2024-2025 season. Read the entire 2024-2025 Respiratory Season Outlook – February Update (2/19/2025)”
- Per Medscape, “A weekly alcohol intake exceeding the limits recommended by US guidelines was associated with an increased risk for coronary heart disease (CHD) among young and middle-aged men and women. Women had a higher risk for CHD than men, especially when heavy episodic drinking was involved.”
- ABC News reports,
- “Rates of drug overdose deaths decreased in the United States for the first time since the COVID-19 pandemic began, according to new federal data published early Thursday.
- “The rate of overdose deaths fell from 32.6 deaths per 100,000 people in 2022 to 31.3 per 100,000 people in 2023, a 4% decrease, according to the report from the Centers for Disease Control and Prevention’s National Center for Health Statistics.
- “Dr. Aitzaz Munir, an assistant professor of psychiatry at Rutgers New Jersey Medical School and associate program director for the Rutgers Addiction Medicine Fellowship Program, told ABC News the drop in the overdose death rate was “surprising” to him but a positive sign.”
- The National Cancer Institute lets us know,
- “Women who are pregnant routinely undergo prenatal blood testing to screen their fetuses for chromosomal disorders such as Down syndrome. However, in rare cases, this noninvasive prenatal testing (NIPT) will result in an abnormal or inconclusive finding that isn’t related to the fetus but rather, to the mother’s DNA.
- “The results of a study of more than 100 women with unusual NIPT findings but a normally developing fetus now show that such findings can have serious implications for the mother. Nearly half of the women in the study turned out to have cancer Exit Disclaimer, the researchers reported December 5 in the New England Journal of Medicine.
- “Most of these cancers were only detected through whole-body magnetic resonance imaging (MRI), which is not yet routinely used by doctors to follow up on abnormal NIPT results. In contrast, standard diagnostic tests, such as physical exams and blood work, missed many of the cancers.
- “These cancers tend to be truly hidden,” said co-lead investigator Amy Turriff, M.S., of the National Human Genome Research Institute. “Our study found that whole-body imaging is critical to sufficiently evaluate women who receive these [abnormal] results for cancer.”
- “I hope [these findings] will increase awareness of these types of results and what should be done to triage these patients to the appropriate levels of care,” said Neeta Vora, M.D., of the University of North Carolina School of Medicine, who studies prenatal genetic testing and maternal cancers but was not involved in the study.”
From the U.S. healthcare business front,
- Beckers Hospital Review identifies the most common reasons for hospitalizations.
- “Maternal and neonatal stays accounted for 22% of hospitalizations in 2021, a recent KFF report found.
- “The report is based on data from the American Hospital Association annual survey, the American Medical Association physician practice benchmark survey, the Census Bureau delineation files and population estimates, the healthcare cost and utilization project national inpatient samples, RAND hospital data and other sources.
- “Maternal stays accounted for 1 in 10 hospitalizations and neonatal stays accounted for the same; hospital stays for mothers and newborns were recorded separately. Medicaid covered about 41% of births nationally.
- “Other hospitalizations were categorized as medical (50%), surgical (18%), injury (5%), and mental health and substance abuse (5%) discharges.”
- Per AIS Health,
- “When it comes to how the country’s three dominant PBMs cover the blockbuster drug Humira (adalimumab) and its many biosimilars, one year has made a major difference.
- “As of 2025, AbbVie’s Humira either “has or will vanish from PBMs’ standard formularies,” Drug Channels CEO Adam Fein, Ph.D., wrote in his annual post analyzing which drugs were excluded on the standard commercial formularies offered by The Cigna Group’s Express Scripts, CVS Health Corp.’s Caremark and UnitedHealth Group’s Optum Rx, as of January 2025.”
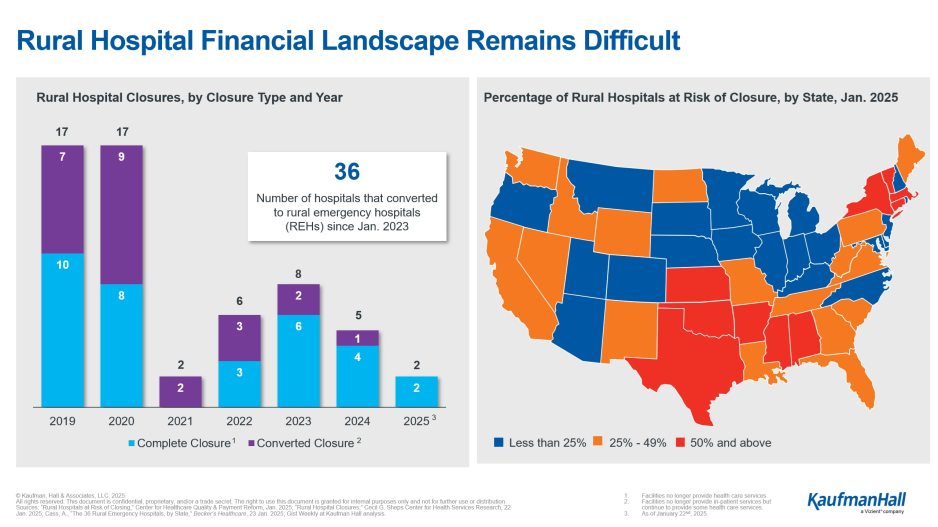
- Kauffman Hall offers an infographic about “The State of Trust in Public Health in AmericaMedCity”
- MedCity News shares seven announcements from the VIVE Conference, a few of which already were included in the FEHBlog.
- Per Fierce Pharma,
- “Pfizer is pulling further away from the gene therapy field with its decision to discontinue hemophilia product Beqvez.
- “The New York pharma is ending global development and commercialization of Beqvez less than a year after an FDA approval for the gene therapy to treat hemophilia B. The one-time treatment carried a list price of $3.5 million per person.
- “Several reasons led to the discontinuation, including limited interest from patients and doctors toward hemophilia gene therapies to date, a Pfizer spokesperson told Fierce Pharma in a statement.
- “No patients seem to have received commercial Beqvez since its FDA nod in April 2024. The Pfizer spokesperson said the company will communicate the news to patients and providers that are in the treatment qualification process, adding that the company remains committed to supporting those who received the med in any clinical trial.
- “Following Beqvez’s exit from the market, Pfizer has no commercial or clinical-stage gene therapies left in the works, according to its website. The spokesperson confirmed that the company doesn’t have any active gene therapy programs at the moment.”