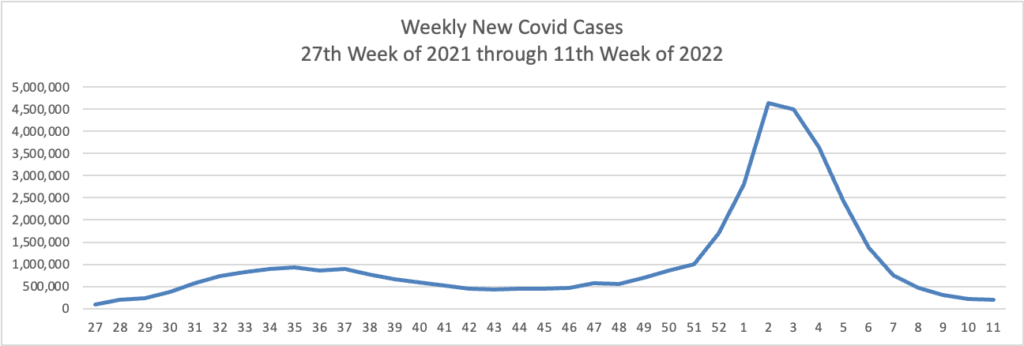
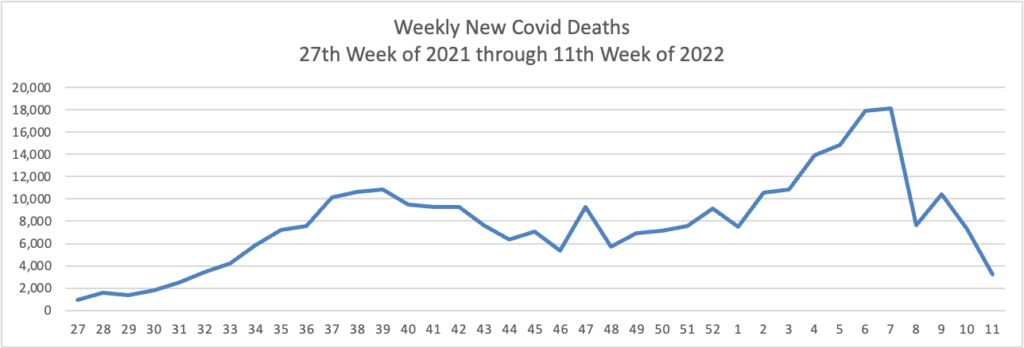
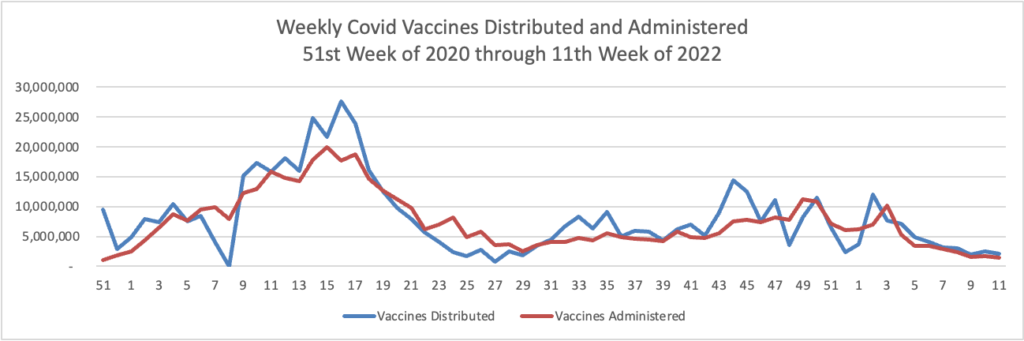
Tuesday’s Tidbits

From the government employment front,
Performance.gov informs us that OPM Director Kiran Ahuja is one of three Leads on implementing Priority One of the President’s Management Agenda — “Strengthen and Empower the Federal Workforce.” Good luck, Director Ahuja.
FedSmith identifies “ten steps federal employees should take at least a decade before retiring to ensure they are prepared to head into their golden years.”
Step 2 of 10 is “Enroll or Stay in FEHB.”
If you plan to continue your federal health benefits in retirement, you must be entitled to retire on an immediate annuity and:
be enrolled for the 5 years immediately before retiring or, during all your federal employment since your first opportunity to enroll if less than 5 years.
Coverage under FEHB, Tricare, or the Civilian Health and Medical Program of Uniformed Services (CHAMPUS) all count toward the 5 year requirement.
In yesterday’s post, the FEHBlog pointed out a Fierce Healthcare interview with the CEO of dialysis heavyweight, Davita. Today, Fierce Healthcare reports
Three big players in kidney care—Fresenius Health Partners, Cricket Health, and InterWell Health—plan to form a new value-based care company focused on services for the earlier stages of kidney disease.
The deal, announced Monday morning, will create a company valued at $2.4 billion, according to the companies.
The merger brings together expertise in value-based kidney care contracting of Fresenius Health Partners, a division of Fresenius Medical Care North America, InterWell Health’s network of more than 1,600 nephrologists and startup Cricket Health’s technology-enabled care model and patient engagement platform. Fresenius Medical Care is the world’s largest operator of dialysis centers.
Healthcare Dive delves into the case for hybrid care models, meaning the marriage of hospital systems and telehealth vendors.
Though the pandemic greatly accelerated the use of telehealth, use of pure-play virtual care has waned as COVID-19 cases drop off across the U.S. But telehealth is increasingly being woven into hospitals’ digital roadmaps, and some are even upping their investments in the modality despite moderating visits, experts said at the annual HIMSS healthcare conference.
That’s giving hope to proponents of hybrid care models, who point to the value of meeting patients where they are to lower costs and improve outcomes.
Intriguing.
Health Payer Intelligence reports
The National Committee for Quality Assurance (NCQA) has awarded [health insurer] Centene the Innovation Award for Health Equity for the company’s Health Equity Improvement model.
“Advancing health equity has a real and positive impact on the health of our members across the country,” said Sarah Bezeredi, senior vice president and chief quality officer for Centene. * * *
At its core, the model uses qualitative and quantitative data to target certain health equity gaps. Centene engaged communities and activated community coalitions in order to empower its strategy. The company’s approach included carefully selecting leaders who would help create and promote the model as well as soliciting community feedback.
In Arizona, Centene sent HbA1c at-home testing kits, which became a common method of chronic disease prevention during the pandemic. The kits were outfitted with a range of delivery modalities to cover the spectrum of members’ needs. Members also received telehealth and telemonitoring tools. The payer incentivized compliance with certain diabetes care measures.
The model tackled patient education in addition to delivering access to care interventions. Centene mailed qualifying members resources about the HbA1c test and comprehensive diabetes care measures.
The payer tracked progress using its Health Equity Dashboard, which leverages HEDIS data and other social determinants of health and demographic data to assess care disparities.
“By using a data-driven process, we are identifying disparity reduction opportunities and tracking performance and success year-over-year,” Bezeredi added.
Mazaal Tov, Centene.