Happy National Doctors’ Day
Today is National Doctors’ Day, and the FEHBlog can think of no better time to thank the medical and para-medical professions for their dedicated and brave service to America and the world.
The FEHBlog learned from reading the Great Influenza that the horrific influenza pandemic in 1918-19 eventually subsided due to a combination of people who survived the disease (and there was a mild wave of the influenza in early 1918) and the enormous number of deaths. As a result, person to person transfer stopped. (Social distancing helped reduce the spread of that illness, e.g., St. Louis, but the FEHBlog is writing about how the pandemic ended.)
NBC News reports that experts believe that people who gain COVID-19 antibodies (though infection) will have immunity for some (at present) unknown period. However, it strikes the FEHBlog, who certainly is not an expert, that a combination of immunized and vaccinated people eventually should defeat the COVID-19 pandemic. The Wall Street Journal reports today that three prescription drug manufacturers are actively seeking to develop a COVID-19 vaccine.
Johnson & Johnson is one of many companies at work on a vaccine for the disease, which has become a global health crisis. Moderna Inc. has begun human trials for a vaccine using a novel approach that relies on the virus’s messenger RNA, a type of genetic material. Sanofi, a French biotech company, has begun work on a similar approach.
Healthcare Dive and Health Payer Intelligence report on additional steps that commercial payers are taking to reduce cost sharing burdens on patients receiving COVID-19 care.
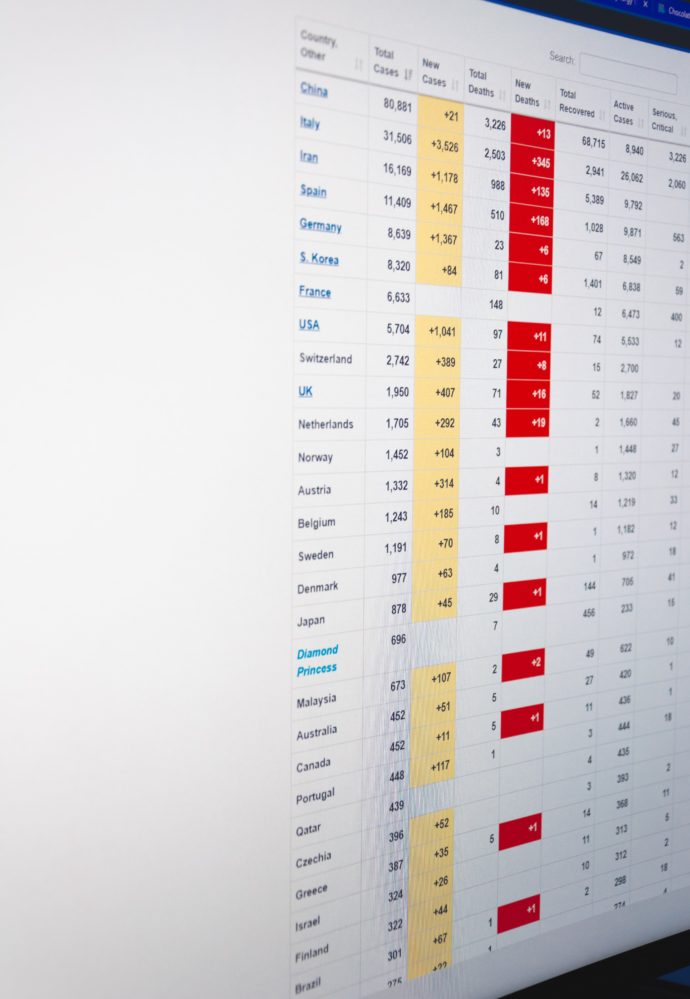
The FEHBlog is confident that due to vastly improved medical care over the past century, Government and private sector support, and public respect for social distancing (which happened in the breach in 1918-19 due in large part to World War 1), the medical and para-medical professions will be able to handle the still rising number of COVID-19 cases while vaccination development continues.