Thursday Miscellany
The Centers for Medicare and Medicaid Services today announced wide ranging Medicare changes to make healthcare more accessible during the COVID-19 emergency. For example,
For the duration of the COVID-19 emergency, CMS is waiving limitations on the types of clinical practitioners that can furnish Medicare telehealth services. Prior to this change, only doctors, nurse practitioners, physician assistants, and certain others could deliver telehealth services. Now, other practitioners are able to provide telehealth services, including physical therapists, occupational therapists, and speech language pathologists.
CMS previously announced that Medicare would pay for certain services conducted by audio-only telephone between beneficiaries and their doctors and other clinicians. Now, CMS is broadening that list to include many behavioral health and patient education services. CMS is also increasing payments for these telephone visits to match payments for similar office and outpatient visits. This would increase payments for these services from a range of about $14-$41 to about $46-$110. The payments are retroactive to March 1, 2020.
It will be interesting to see whether commercial plans adopt these changes (perhaps they already have). Check out the lengthy list. Hopefully many of these changes will be made permanent following the COVID-19 emergency.
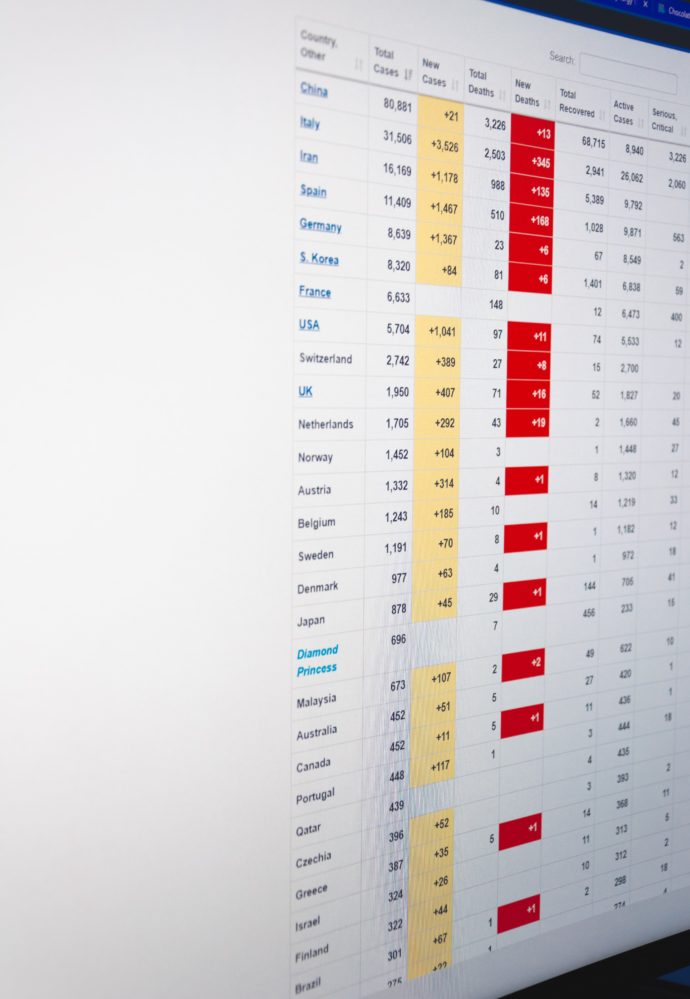
As the FEHBlog has listed major COVID-19 testing locations. It occurred that he should also link to the COVID-19 sites for the major actuarial consulting firms, all of which provide useful information for health plans:
These sites are a valuable public service in the FEHBlog’s view.
HHS’s Office for Civil Rights today provided a helpful COVID-19 cyber threat resources. While the FEHBlog could not find a link to the list, he was able to upload the email to Dropbox.