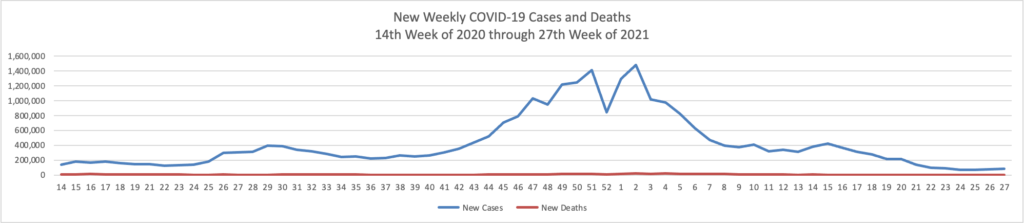
Based on the Centers for Disease Control’s COVID-19 Data Tracker website, here is the FEHBlog’s chart of new weekly COVID-19 cases and deaths over the 14th week of 2020 through 27th week of this year (beginning April 2, 2020, and ending July 7, 2021; using Thursday as the first day of the week in order to facilitate this weekly update):
and here is the CDC’s latest overall weekly hospitalization rate chart for COVID-19:

The FEHBlog has noticed that the new cases and deaths chart shows a flat line for new weekly deaths because new cases significantly exceed new deaths. Accordingly here is a chart of new COVID-19 deaths over the period (April 2, 2020, through July 7, 2021):
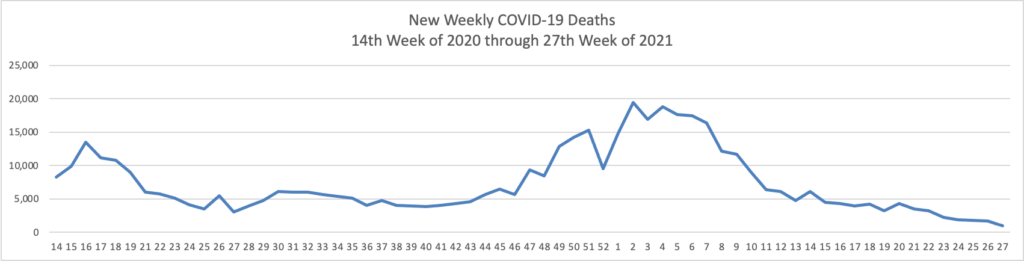
This is the first week since the FEHBlog began the chart that the number of new weekly deaths (955) has fallen below 1,000 nationwide.
Finally here is a COVID-19 vaccinations chart over the period December 17, 2020, through July 7, 2021 which also uses Thursday as the first day of the week:
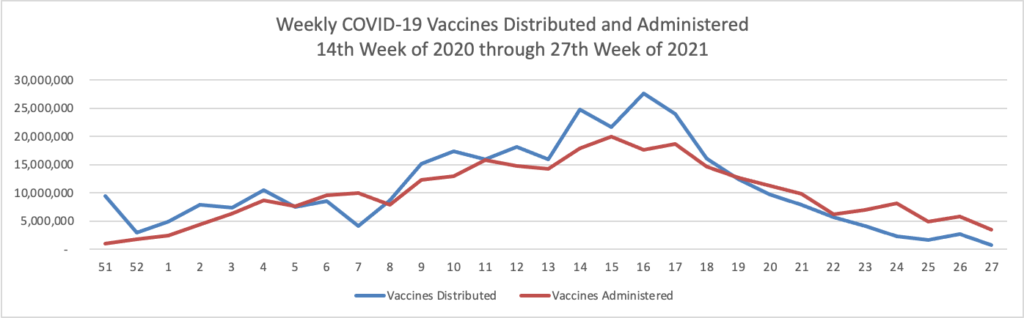
The Centers for Disease Control reports that “The COVID Data Tracker Vaccination Demographic Trends tab shows vaccination trends by age group. As of July 8, 88.5% of people ages 65 or older have received at least one dose of vaccine and 78.8% are fully vaccinated. Just over two-thirds (67.3%) of people ages 18 or older have received at least one dose of vaccine and 58.5% are fully vaccinated. For people ages 12 or older, 64.5% have received at least one dose of vaccine and 55.8% are fully vaccinated.”
Today, the President issued a wide ranging executive order on promoting competition in the American economy, which of course had been humming along nicely before the pandemic struck. Here’s a link to the fact sheet on the executive order which recounts its 70+ initiatives. The FEHBlog call attention to several healthcare initiatives in Sunday’s posts. The FEHBlog sense that this executive order will generate a lot of litigation.
The Wall Street Journal reports that
The Food and Drug Administration’s acting commissioner, Janet Woodcock, is taking the highly unusual step of asking for a federal investigation of doctors within her own agency who met with the makers of an Alzheimer’s drug before the medicine’s recent approval.
Dr. Woodcock, in a letter made public Friday, called for the Office of Inspector General of the Department of Health and Human Services, which oversees the FDA, to review interactions between the drugmaker [Biogen] and FDA staff during the approval process.
The drug’s approval has been highly controversial, partly because of its annual price pegged at $56,000, and partly because evidence of the drug’s effectiveness was inconclusive.
This unusual step certainly will make the FDA decision makers think twice before crossing their advisory committee again. Biogen added fuel to the fire with its outrageous pricing.
